Which Of The Following Is The Strongest Acid
Holbox
Mar 31, 2025 · 5 min read
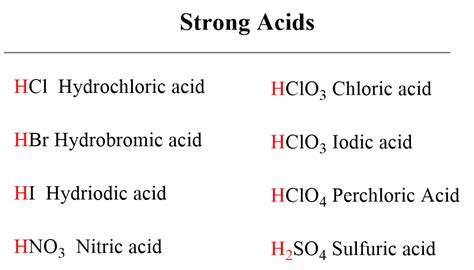
Table of Contents
- Which Of The Following Is The Strongest Acid
- Table of Contents
- Which of the Following is the Strongest Acid? Understanding Acid Strength and pKa Values
- What Makes an Acid Strong?
- Introducing pKa: A More Precise Measure of Acid Strength
- Factors Affecting Acid Strength
- Comparing Acids: A Practical Example
- Beyond pKa: Other Considerations
- Conclusion: A Multifaceted Understanding
- Latest Posts
- Latest Posts
- Related Post
Which of the Following is the Strongest Acid? Understanding Acid Strength and pKa Values
Determining the strongest acid from a given set requires a deep understanding of acid strength and the factors that influence it. While simply comparing pH values might seem like a straightforward approach, it’s crucial to consider the context and, more importantly, the concept of pKa. This article delves into the intricacies of acid strength, explaining the significance of pKa values and providing a framework for identifying the strongest acid among a group of contenders. We'll explore various acid types, the factors impacting their strength, and illustrate the concept with examples.
What Makes an Acid Strong?
The strength of an acid is determined by its ability to donate a proton (H⁺) to a base. Strong acids completely dissociate in water, meaning they donate all their protons to water molecules. Weak acids, conversely, only partially dissociate. The equilibrium between the undissociated acid (HA) and its conjugate base (A⁻) and H⁺ ions dictates the acid's strength.
The equilibrium reaction for a generic acid (HA) in water is:
HA(aq) + H₂O(l) ⇌ H₃O⁺(aq) + A⁻(aq)
A higher concentration of H₃O⁺ (hydronium ions) signifies a stronger acid because more protons are donated.
Introducing pKa: A More Precise Measure of Acid Strength
While pH measures the concentration of H⁺ ions in a solution at a given time, it doesn't inherently reflect the intrinsic strength of an acid. pH can change based on concentration. pKa, on the other hand, is a constant that directly reflects the acid's inherent strength independent of concentration. It’s the negative logarithm (base 10) of the acid dissociation constant (Ka):
pKa = -log₁₀(Ka)
A lower pKa value indicates a stronger acid. A lower pKa means a higher Ka, signifying a greater tendency for the acid to donate its proton and achieve equilibrium with a higher concentration of H₃O⁺.
Factors Affecting Acid Strength
Several factors influence an acid's strength and consequently its pKa value:
-
Electronegativity: The electronegativity of the atom bonded to the acidic hydrogen plays a critical role. A more electronegative atom attracts electrons more strongly, weakening the O-H bond and making proton donation easier. This leads to a stronger acid. For example, the increasing electronegativity across the halogens (F > Cl > Br > I) results in increasing acid strength for the hydrohalic acids (HF, HCl, HBr, HI). However, HF is an exception due to the strong H-F bond.
-
Inductive Effect: Electron-withdrawing groups attached to the molecule can stabilize the conjugate base, making the acid stronger. These groups pull electron density away from the acidic hydrogen, facilitating proton release.
-
Resonance: If the conjugate base can be stabilized through resonance, the acid will be stronger. Resonance delocalizes the negative charge, making the conjugate base more stable and therefore more likely to form.
-
Hybridization: The hybridization of the atom bearing the acidic hydrogen also affects the acid strength. A higher s-character in the hybrid orbital leads to a stronger acid. For example, sp hybridized carbons are more acidic than sp³ hybridized carbons.
-
Size and Polarizability: Larger atoms are more polarizable, meaning their electron clouds are more easily distorted. This can lead to weaker bonds and stronger acids, as seen in the trend of hydrohalic acid strengths (HI > HBr > HCl).
Comparing Acids: A Practical Example
Let's consider the following acids:
- Hydrochloric acid (HCl): A strong mineral acid with a pKa of approximately -7.
- Acetic acid (CH₃COOH): A weak organic acid with a pKa of approximately 4.76.
- Hydrofluoric acid (HF): A weak acid with a pKa of approximately 3.2.
- Sulfuric acid (H₂SO₄): A strong diprotic acid. The first proton dissociation has a pKa of approximately -3, the second is weaker.
- Benzoic acid (C₆H₅COOH): A weak organic acid with a pKa of approximately 4.2.
Based on their pKa values, the order of acid strength from strongest to weakest is:
HCl > H₂SO₄ > HF > Benzoic acid > Acetic acid
HCl is the strongest acid in this list due to its significantly lower pKa value. Its complete dissociation in water leads to a high concentration of H₃O⁺ ions. Sulfuric acid is a strong acid but slightly weaker than HCl. While HF's pKa is higher than HCl and H₂SO₄, it's still relatively strong compared to carboxylic acids like acetic and benzoic acid. The difference in strength between benzoic acid and acetic acid is relatively small. Benzoic acid is slightly stronger due to the electron-withdrawing effect of the phenyl group.
Beyond pKa: Other Considerations
While pKa is a powerful tool for comparing acid strengths, it’s crucial to consider the context. The actual pH of a solution depends on both the pKa of the acid and its concentration. A highly concentrated solution of a weak acid can have a lower pH than a dilute solution of a strong acid.
Furthermore, the solvent also plays a role. Acid strengths can vary considerably in different solvents. Water is the standard solvent for comparing pKa values, but the values can change dramatically in non-aqueous solvents.
Conclusion: A Multifaceted Understanding
Determining the strongest acid from a group requires more than just a superficial glance. The pKa value is the most reliable indicator of an acid's intrinsic strength. By considering factors like electronegativity, inductive effect, resonance, hybridization, and size/polarizability, we can gain a deeper understanding of how these factors influence an acid's ability to donate protons. This multifaceted perspective allows for accurate comparisons and predictions of acid strengths in diverse chemical scenarios. Remember to always consider the context and potential influence of concentration and solvent when interpreting acid strength data. Understanding pKa is fundamental to comprehending and predicting chemical reactivity in numerous fields, from organic chemistry to biochemistry and environmental science.
Latest Posts
Latest Posts
-
How Could Twitter Maximize Its Effectiveness
Apr 03, 2025
-
Is An Example Of Impossible Foods Using Product Placement
Apr 03, 2025
-
Which Of The Following Statements About Diabetes Mellitus Is False
Apr 03, 2025
-
Nrp 8th Edition Instructor Maintenance Requirements
Apr 03, 2025
-
The Thickness Of A Flange On An Aircraft Component
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Is The Strongest Acid . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
