Which Of The Following Is A Radical Scavenger
Holbox
Mar 31, 2025 · 6 min read
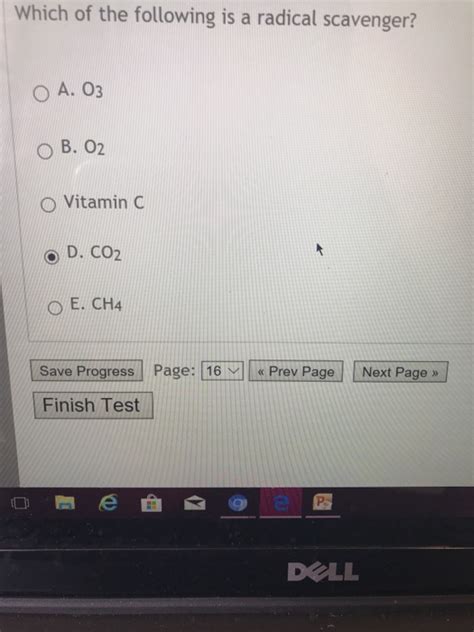
Table of Contents
- Which Of The Following Is A Radical Scavenger
- Table of Contents
- Which of the Following is a Radical Scavenger? Understanding Antioxidant Power
- What are Free Radicals?
- The Role of Radical Scavengers (Antioxidants)
- Identifying Radical Scavengers: Key Characteristics
- Examples of Radical Scavengers: A Diverse Group
- Mechanisms of Action: How Radical Scavengers Work
- Measuring Radical Scavenging Activity: Laboratory Techniques
- The Importance of Dietary Antioxidants
- Conclusion: A Complex Network of Protection
- Latest Posts
- Latest Posts
- Related Post
Which of the Following is a Radical Scavenger? Understanding Antioxidant Power
Free radicals, unstable molecules with unpaired electrons, are a natural byproduct of cellular metabolism. While our bodies produce antioxidants to neutralize these damaging molecules, an excess of free radicals can lead to oxidative stress, contributing to various health problems. This article delves into the world of free radicals and radical scavengers, explaining what they are, how they interact, and which compounds exhibit this crucial antioxidant property. We'll explore numerous examples and examine the mechanisms behind their effectiveness.
What are Free Radicals?
Free radicals are highly reactive molecules that readily interact with other molecules, often damaging them in the process. This reactivity stems from their unpaired electron, which makes them highly unstable and eager to achieve a stable state. This often involves stealing an electron from another molecule, thereby creating a new free radical and perpetuating a chain reaction of damage. This chain reaction can affect various biomolecules including:
- Lipids: Leading to lipid peroxidation and membrane damage.
- Proteins: Causing protein oxidation and loss of function.
- DNA: Resulting in DNA mutations and potentially cancer.
The Role of Radical Scavengers (Antioxidants)
Radical scavengers, also known as antioxidants, are substances that can neutralize free radicals by donating an electron without becoming a free radical themselves. This effectively stops the chain reaction of oxidative damage. They achieve this neutralization through various mechanisms, including:
- Hydrogen Atom Transfer (HAT): The antioxidant donates a hydrogen atom to the free radical, stabilizing it.
- Single Electron Transfer (SET): The antioxidant donates a single electron to the free radical, forming a stable radical cation and a stable anion.
- Radical Adduct Formation: The antioxidant reacts with the free radical to form a stable adduct.
Identifying Radical Scavengers: Key Characteristics
Identifying a potential radical scavenger requires understanding its chemical properties and its ability to effectively donate electrons without generating further instability. Several characteristics suggest potent radical scavenging activity:
-
Presence of phenolic hydroxyl groups: Compounds containing multiple phenolic hydroxyl groups (-OH) are particularly effective at donating electrons. These groups are readily oxidized, meaning they easily give up an electron to neutralize a free radical.
-
Conjugated double bonds: A system of conjugated double bonds creates electron delocalization, enhancing the molecule's stability even after electron donation. This stabilizes the antioxidant after it donates an electron, preventing it from becoming a new free radical.
-
Electron-donating substituents: The presence of electron-donating groups (like methyl or methoxy groups) on the aromatic ring further enhances the antioxidant's ability to donate electrons.
-
Resonance stabilization: Molecules with extensive resonance structures are more stable, reducing the likelihood of them initiating further radical reactions after donating an electron.
Examples of Radical Scavengers: A Diverse Group
A wide range of compounds exhibit radical scavenging activity. These include:
-
Vitamin C (Ascorbic Acid): A potent water-soluble antioxidant with multiple hydroxyl groups capable of donating electrons to neutralize free radicals. It's crucial for protecting against oxidative stress in the body's aqueous environments.
-
Vitamin E (Tocopherols): A fat-soluble antioxidant with a phenolic structure. It effectively protects cell membranes from lipid peroxidation by neutralizing free radicals in lipid-rich environments.
-
Beta-Carotene: A carotenoid with a highly conjugated double bond system, providing strong radical scavenging ability. Its effectiveness in protecting against oxidative stress is attributed to its ability to quench singlet oxygen, a highly reactive form of oxygen.
-
Glutathione: An important intracellular antioxidant, this tripeptide plays a vital role in protecting cells against oxidative damage. Its thiol group (-SH) is responsible for its antioxidant properties.
-
Coenzyme Q10 (Ubiquinone): A lipid-soluble antioxidant found in the mitochondria, crucial for energy production. It plays a significant role in protecting mitochondrial membranes from oxidative damage.
-
Polyphenols: A large class of compounds found in various plants. Many polyphenols, like those found in green tea (catechins), berries (anthocyanins), and grapes (resveratrol), possess potent antioxidant activity due to their multiple hydroxyl groups and conjugated double bond systems.
-
Flavonoids: Subgroup of polyphenols known for their significant antioxidant activity. Various flavonoids, including quercetin, rutin, and hesperidin, exhibit potent radical scavenging properties.
-
Selenium: This essential trace mineral is a component of the enzyme glutathione peroxidase, which plays a critical role in protecting cells from oxidative damage. Selenium helps to reduce the damaging effects of hydrogen peroxide and other reactive oxygen species.
Mechanisms of Action: How Radical Scavengers Work
The specific mechanisms by which different radical scavengers neutralize free radicals vary, but they all share the common theme of donating electrons or hydrogen atoms to stabilize the free radical without creating a new one. This process effectively interrupts the chain reaction of oxidative damage. The specific mechanism often depends on the chemical structure of the scavenger and the type of free radical being neutralized.
Measuring Radical Scavenging Activity: Laboratory Techniques
Scientists use various laboratory techniques to measure the radical scavenging activity of different compounds. Common methods include:
-
DPPH (2,2-diphenyl-1-picrylhydrazyl) Assay: This assay measures the ability of a compound to reduce the DPPH radical, a stable nitrogen-centered free radical. The decrease in DPPH absorbance indicates the radical scavenging activity of the tested compound.
-
ABTS (2,2'-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid)) Assay: Similar to the DPPH assay, this method measures the ability of a compound to reduce the ABTS radical cation. The decrease in ABTS absorbance is directly proportional to the radical scavenging activity.
-
FRAP (Ferric Reducing Antioxidant Power) Assay: This assay measures the ability of a compound to reduce ferric ions (Fe3+) to ferrous ions (Fe2+). The reducing power indicates the antioxidant capacity of the compound.
-
Oxygen Radical Absorbance Capacity (ORAC) Assay: This assay measures the ability of a compound to inhibit the oxidation of a fluorescent probe by free radicals. The higher the inhibition, the greater the antioxidant capacity.
The Importance of Dietary Antioxidants
A diet rich in fruits, vegetables, and other plant-based foods provides a plentiful source of natural radical scavengers. This intake contributes significantly to protecting the body from oxidative stress and its associated health risks.
Conclusion: A Complex Network of Protection
The question "Which of the following is a radical scavenger?" doesn't have a single answer; many compounds demonstrate this crucial antioxidant property. The effectiveness of a radical scavenger depends on various factors, including its chemical structure, the type of free radical being neutralized, and the specific environment. Understanding the role of free radicals and radical scavengers is crucial for maintaining health and preventing various diseases linked to oxidative stress. A balanced diet rich in antioxidant-rich foods, along with a healthy lifestyle, contributes to optimal protection against the damaging effects of free radicals. Further research continues to reveal the intricate mechanisms and the wide diversity of compounds possessing this vital protective capacity.
Latest Posts
Latest Posts
-
H C C H Lewis Structure
Apr 03, 2025
-
The Image Shows A Top Down View Of A Wind Turbine
Apr 03, 2025
-
Nutrition Through The Life Cycle 7th Edition
Apr 03, 2025
-
Add Substituents To Draw The Conformer Below
Apr 03, 2025
-
A Firm Pursuing A Best Cost Provider Strategy
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Is A Radical Scavenger . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
