For A Review Of How To Make Alkyl Tosylates
Holbox
Mar 31, 2025 · 5 min read
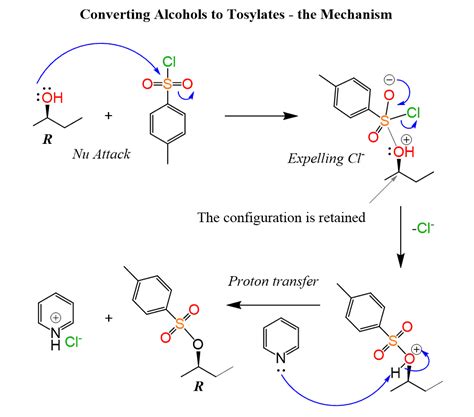
Table of Contents
- For A Review Of How To Make Alkyl Tosylates
- Table of Contents
- A Comprehensive Review of Alkyl Tosylate Synthesis
- Understanding the Tosylate Group and its Reactivity
- Synthesis Methods: A Detailed Exploration
- 1. Reaction of Alcohols with p-Toluenesulfonyl Chloride (TsCl)
- 2. Considerations for Sterically Hindered Alcohols
- 3. Protecting Group Strategies
- 4. Purification Techniques
- Applications of Alkyl Tosylates in Organic Synthesis
- 1. Nucleophilic Substitution Reactions (SN1 and SN2)
- 2. Elimination Reactions
- 3. Other Reactions
- Advantages and Disadvantages of using Alkyl Tosylates
- Safety Precautions
- Conclusion
- Latest Posts
- Latest Posts
- Related Post
A Comprehensive Review of Alkyl Tosylate Synthesis
Alkyl tosylates, also known as alkyl p-toluenesulfonates, are valuable intermediates in organic synthesis. Their versatility stems from the excellent leaving group ability of the tosylate group, making them readily susceptible to nucleophilic substitution reactions, eliminations, and other transformations. This review delves into the various methods for synthesizing alkyl tosylates, exploring their mechanisms, advantages, limitations, and practical considerations. We’ll examine the choice of reagents, reaction conditions, and purification techniques, providing a comprehensive guide for organic chemists of all levels.
Understanding the Tosylate Group and its Reactivity
The tosylate group (-OTs) is derived from p-toluenesulfonic acid (TsOH), a strong organic acid. The sulfonate ester linkage incorporates a resonance-stabilized anion, making it an exceptionally good leaving group. This characteristic is crucial for the synthetic utility of alkyl tosylates. The electron-withdrawing nature of the p-toluenesulfonyl group (-Ts) stabilizes the negative charge that develops during the departure of the tosylate anion, facilitating nucleophilic substitution reactions. This superior leaving group ability compared to halides like chloride or bromide allows for reactions to proceed under milder conditions.
Synthesis Methods: A Detailed Exploration
Several methods are available for preparing alkyl tosylates, each with its own advantages and limitations. The most common approach involves the reaction of an alcohol with p-toluenesulfonyl chloride (TsCl) in the presence of a base.
1. Reaction of Alcohols with p-Toluenesulfonyl Chloride (TsCl)
This is the most widely used and versatile method for alkyl tosylate synthesis. The reaction typically proceeds via an SN2 mechanism:
Reaction: ROH + TsCl + Base → ROTs + HCl + Base-H<sup>+</sup>
Mechanism:
- Nucleophilic Attack: The alcohol's oxygen atom acts as a nucleophile, attacking the sulfur atom of TsCl. This forms a tetrahedral intermediate.
- Proton Transfer: A base (e.g., pyridine, triethylamine) abstracts a proton from the hydroxyl group of the tetrahedral intermediate.
- Elimination: The chloride ion leaves, generating the alkyl tosylate and liberating HCl. The base neutralizes the HCl formed.
Choice of Base: The choice of base significantly influences the reaction's efficiency. Pyridine is a commonly used base due to its relatively low nucleophilicity, minimizing the risk of competing reactions. However, other bases like triethylamine or N,N-diisopropylethylamine (DIPEA) can be employed, especially for sterically hindered alcohols.
Solvent Selection: The solvent should be inert towards both the alcohol and TsCl. Common solvents include dichloromethane (DCM), chloroform, and diethyl ether. The choice of solvent may depend on the solubility of the reactants and the desired reaction rate.
2. Considerations for Sterically Hindered Alcohols
Sterically hindered alcohols may react sluggishly or not at all with TsCl under standard conditions. In such cases, stronger bases and/or elevated temperatures might be necessary. However, it's crucial to carefully monitor the reaction conditions to prevent side reactions such as elimination. Sometimes, alternative approaches involving different leaving groups might be considered.
3. Protecting Group Strategies
Alkyl tosylates can serve as excellent protecting groups for alcohols. This is particularly useful in multi-step syntheses where selective protection is crucial. The tosylate group can be easily introduced and removed under specific conditions, allowing for selective manipulation of other functional groups.
4. Purification Techniques
After the reaction is complete, the alkyl tosylate needs to be purified. Common purification techniques include:
- Extraction: The alkyl tosylate can be extracted from the reaction mixture using an appropriate solvent.
- Recrystallization: This is effective for purifying solid alkyl tosylates.
- Chromatography: Column chromatography or flash chromatography may be required for complex reaction mixtures.
Important Note: Alkyl tosylates are often sensitive to moisture and should be handled under anhydrous conditions. Prolonged exposure to moisture may lead to hydrolysis and the regeneration of the alcohol.
Applications of Alkyl Tosylates in Organic Synthesis
The superior leaving group ability of the tosylate renders alkyl tosylates highly versatile intermediates in a wide range of organic transformations:
1. Nucleophilic Substitution Reactions (SN1 and SN2)
Alkyl tosylates readily participate in both SN1 and SN2 reactions. The choice of nucleophile and reaction conditions will dictate whether the reaction proceeds via an SN1 or SN2 mechanism. Examples include:
- Synthesis of ethers: Reaction with alkoxides yields ethers.
- Synthesis of amines: Reaction with amines gives substituted amines.
- Synthesis of thioethers: Reaction with thiols generates thioethers.
- Synthesis of nitriles: Reaction with cyanide ions results in the formation of nitriles.
2. Elimination Reactions
Under appropriate conditions, alkyl tosylates can undergo elimination reactions to form alkenes. This usually requires a strong base and elevated temperatures. The type of base and reaction conditions (temperature, solvent) influence whether E1 or E2 elimination is favored.
3. Other Reactions
Alkyl tosylates can also participate in other reactions, including:
- Grignard Reactions: Formation of Grignard reagents from alkyl tosylates via reduction followed by reaction with magnesium.
- Reduction: Reduction of alkyl tosylates using reducing agents like lithium aluminum hydride (LiAlH4) to yield the corresponding alkane.
Advantages and Disadvantages of using Alkyl Tosylates
Advantages:
- Excellent leaving group: Facilitates reactions under milder conditions.
- Versatile reactivity: Participates in various transformations (SN1, SN2, elimination).
- Crystalline nature (often): Facilitates purification and handling.
- Selective protection: Allows for selective functional group manipulation.
Disadvantages:
- Sensitivity to moisture: Requires anhydrous conditions.
- Potential for side reactions: Elimination reactions may compete with substitution, particularly with sterically hindered substrates.
- Preparation may require multiple steps: Depending on the starting material.
Safety Precautions
- p-Toluenesulfonyl chloride (TsCl) is a corrosive and irritating substance. Handle it with appropriate safety precautions, including gloves, eye protection, and a well-ventilated area.
- Alkyl tosylates should be handled under anhydrous conditions to prevent hydrolysis.
- Dispose of waste materials properly according to local regulations.
Conclusion
Alkyl tosylates are indispensable intermediates in organic synthesis due to their excellent leaving group ability and versatile reactivity. The synthesis of alkyl tosylates, primarily through the reaction of alcohols with TsCl, is a well-established and widely utilized procedure. A careful consideration of the choice of reagents, reaction conditions, and purification techniques is vital for achieving high yields and minimizing side reactions. This comprehensive review highlights the critical aspects of alkyl tosylate synthesis and applications, equipping organic chemists with the knowledge to effectively utilize these versatile building blocks in their synthetic endeavors. Careful attention to safety protocols is paramount throughout the process.
Latest Posts
Latest Posts
-
What Is The Patient Care Partnership
Apr 03, 2025
-
Consider The Circuit Diagram In The Figure
Apr 03, 2025
-
If The Following Items Needed To Be Stored
Apr 03, 2025
-
Comparative Advantage Refers To The Ability To
Apr 03, 2025
-
One Of The Dangers Of Self Report Personality Measures Is That
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about For A Review Of How To Make Alkyl Tosylates . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
