Draw Both The Organic And Inorganic Intermediate Species
Holbox
Mar 31, 2025 · 6 min read
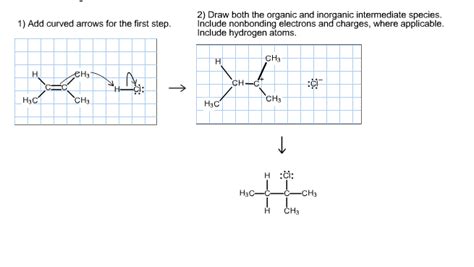
Table of Contents
- Draw Both The Organic And Inorganic Intermediate Species
- Table of Contents
- Drawing Organic and Inorganic Intermediate Species: A Comprehensive Guide
- What are Intermediate Species?
- Drawing Organic Intermediates: A Step-by-Step Approach
- Drawing Inorganic Intermediates: A Diverse Landscape
- Importance of Drawing Intermediates
- Advanced Techniques and Considerations
- Latest Posts
- Latest Posts
- Related Post
Drawing Organic and Inorganic Intermediate Species: A Comprehensive Guide
Understanding reaction mechanisms requires visualizing the intermediate species formed during the transformation of reactants into products. This skill is crucial for organic and inorganic chemistry, and proficiency in drawing these intermediates is essential for success in these fields. This article will provide a comprehensive guide to drawing both organic and inorganic intermediate species, covering various types and illustrating with examples.
What are Intermediate Species?
Intermediate species are transient molecules or ions that exist only briefly during a chemical reaction. They are neither reactants nor products but crucial stepping stones in the reaction pathway. They are typically short-lived, highly reactive, and difficult to isolate. Their existence is often inferred from experimental observations and theoretical calculations.
Drawing Organic Intermediates: A Step-by-Step Approach
Organic intermediates are characterized by their carbon-based structures and often involve the participation of electrons in bond formation and breaking. Common types include:
1. Carbocations:
- Definition: Positively charged carbon atoms with only three bonds. They are highly electrophilic and readily react with nucleophiles.
- Drawing: Represent the positive charge using a "+" symbol directly above the carbon atom. Remember to show the three bonds to the carbon.
- Example: A tertiary carbocation (most stable) would be depicted as:
(C)C
2. Carbanions:
- Definition: Negatively charged carbon atoms with three bonds and a lone pair of electrons. They are nucleophilic and react with electrophiles.
- Drawing: Show the negative charge using a "-" symbol directly above the carbon atom and include the three bonds and lone pair.
- Example: A primary carbanion would be depicted as:
[C⁻]CC
3. Radicals:
- Definition: Atoms or molecules with an unpaired electron, making them highly reactive.
- Drawing: Represent the unpaired electron using a single dot (•) next to the atom possessing it.
- Example: A methyl radical would be drawn as:
•CH₃
4. Carbenes:
- Definition: Neutral molecules containing a divalent carbon atom with two bonds and two non-bonding electrons.
- Drawing: Depict the two non-bonding electrons as two dots next to the carbon atom. Show the two bonds to the carbon.
- Example: Methylene carbene would be:
:CH₂
5. Transition States:
- Definition: High-energy species representing the peak of the energy barrier in a reaction. They are not true intermediates but crucial points on the reaction coordinate.
- Drawing: Transition states are typically represented using a double dagger symbol (‡) and are often depicted with partial bonds using dashed lines to illustrate bond formation or breaking. This is a more complex representation often requiring a deeper understanding of the reaction mechanism.
- Example: A transition state involving bond formation could be schematically shown using dashed lines representing partial bonds.
Illustrative Examples of Organic Intermediate Mechanisms:
Let's illustrate these intermediates within reaction mechanisms. Consider the SN1 reaction (Substitution Nucleophilic Unimolecular) where a tertiary alkyl halide reacts with a nucleophile:
-
Step 1: Formation of a carbocation: The alkyl halide undergoes ionization, leading to the formation of a carbocation intermediate. This step is rate-determining. The carbocation would be drawn as described above.
-
Step 2: Nucleophilic attack: The nucleophile attacks the carbocation, forming a new bond and resulting in the product.
Another example is the electrophilic addition of HBr to an alkene. The reaction proceeds through a carbocation intermediate:
-
Step 1: Protonation of the alkene: A proton adds to the alkene, forming a carbocation.
-
Step 2: Nucleophilic attack by bromide: The bromide ion attacks the carbocation, forming the final product.
Drawing these intermediates accurately helps in understanding the flow of electrons and the stereochemical aspects of the reaction.
Drawing Inorganic Intermediates: A Diverse Landscape
Inorganic intermediates exhibit greater diversity in structure and bonding compared to their organic counterparts. They can involve various elements and bonding patterns, including coordination complexes, radicals, and ions with varying charges.
1. Metal Complexes with Variable Oxidation States: Many inorganic reactions involve changes in the oxidation state of a metal ion. The intermediate species often include complexes with unusual coordination numbers or geometries.
- Drawing: Use standard coordination chemistry notations, showing the metal ion at the center surrounded by ligands. Indicate oxidation states using Roman numerals.
- Example: A square planar complex of Pd(II) with two chloride and two ammonia ligands would be [PdCl₂(NH₃)₂].
2. Radicals and Reactive Intermediates: Inorganic radicals, analogous to organic radicals, play a significant role in numerous reactions. These species contain unpaired electrons and are highly reactive.
- Drawing: Similar to organic radicals, an unpaired electron is represented by a single dot near the atom carrying it.
- Example: A chlorine radical would be represented as ·Cl.
3. Ions with Varying Charges: Many inorganic reactions involve ions with varying charges. Drawing these requires careful attention to the charge and the surrounding atoms.
- Drawing: Clearly indicate the charge on the ion using appropriate superscripts.
- Example: A sulfate ion would be SO₄²⁻.
4. Bridging Ligands: Bridging ligands connect two or more metal centers in coordination complexes. Their representation requires careful attention to the connectivity and coordination modes.
- Drawing: Draw the ligands connecting the metal centers appropriately.
- Example: A bridging hydroxide ligand (µ-OH) connects two metal ions.
Illustrative Examples of Inorganic Reaction Mechanisms:
Consider the oxidation of iodide by hydrogen peroxide catalyzed by iron(II) ions. This reaction involves a series of steps with several intermediates:
-
Fe²⁺ + H₂O₂ → Fe³⁺ + HO· + OH⁻: The iron(II) ion reduces hydrogen peroxide generating a hydroxyl radical (HO·).
-
Fe²⁺ + HO· → Fe³⁺ + OH⁻: The hydroxyl radical is further reduced by iron(II).
-
2 I⁻ + 2 HO· → I₂ + 2 OH⁻: The hydroxyl radicals oxidize iodide to iodine.
The mechanism involves several intermediate species, including the hydroxyl radical (HO·) and iron(III) ions (Fe³⁺). Drawing these intermediates is vital for understanding the redox processes.
Another example involves the formation of a coordination complex: The reaction of a metal ion with ligands involves the formation of intermediate complexes with varying coordination numbers.
Importance of Drawing Intermediates
Accurately drawing intermediates is critical for several reasons:
-
Understanding reaction mechanisms: Visualizing the intermediates provides insights into the stepwise nature of the reaction.
-
Predicting reaction products: Knowing the structure and reactivity of intermediates helps in predicting the products of a chemical reaction.
-
Developing synthetic strategies: Understanding reaction mechanisms and intermediate species is essential for designing new synthetic routes for the preparation of various molecules.
-
Interpreting experimental data: Observed experimental data, such as kinetic studies, often support the existence and nature of postulated intermediates.
-
Computational chemistry: Theoretical calculations often involve studying the structure and energy of intermediate species to validate proposed mechanisms.
Advanced Techniques and Considerations
As you progress, more sophisticated techniques become important:
-
Resonance structures: Many intermediates exhibit resonance, meaning they can be represented by multiple Lewis structures. Drawing all significant resonance structures is important to accurately capture the electron distribution.
-
Stereochemistry: Consider stereochemistry when drawing intermediates. Chiral centers may be created or destroyed during the reaction, significantly influencing the stereochemistry of the products.
-
3D representations: For complex intermediates, using 3D models or software can enhance visualization and understanding of their spatial arrangements.
Mastering the art of drawing organic and inorganic intermediates is a crucial skill in chemistry. Consistent practice, careful attention to detail, and a firm understanding of chemical principles are key to success. By diligently studying examples and applying the techniques outlined above, you can develop the proficiency necessary to confidently visualize and depict these essential components of chemical reactions. Remember that this is a skill that develops over time and consistent effort will yield significant improvements.
Latest Posts
Latest Posts
-
Daily Capacity Of A Product Layout Is Determined By
Apr 05, 2025
-
Usually Returns A Body Part To Zero Position
Apr 05, 2025
-
Suppose The Canadian Government Has Decided
Apr 05, 2025
-
If The Required Adjusting Entry For Accrued Salaries Is Omitted
Apr 05, 2025
-
Which Statement About Pay Ranges Is Most Accurate
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Draw Both The Organic And Inorganic Intermediate Species . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
