Choose The Best Lewis Structure For Xei2
Holbox
Mar 31, 2025 · 5 min read
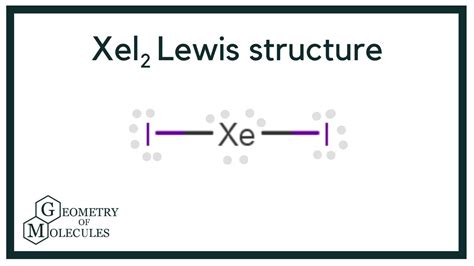
Table of Contents
- Choose The Best Lewis Structure For Xei2
- Table of Contents
- Choosing the Best Lewis Structure for XeI₂: A Comprehensive Guide
- Understanding Lewis Structures and the Octet Rule
- Possible Lewis Structures for XeI₂
- Structure 1: Linear Structure with Expanded Octet on Xenon
- Structure 2: Bent Structure with Expanded Octet on Xenon
- Structure 3: Structures Violating the Octet Rule (less likely)
- Evaluating the Structures: Choosing the Best Lewis Structure
- Why the Linear Structure (Structure 1) is the Best
- Deeper Dive into VSEPR Theory and XeI₂ Geometry
- Applications and Further Considerations
- Conclusion: The Importance of a Rigorous Approach
- Latest Posts
- Latest Posts
- Related Post
Choosing the Best Lewis Structure for XeI₂: A Comprehensive Guide
The noble gas xenon (Xe) defies the traditional octet rule, readily forming compounds like XeI₂. Determining the best Lewis structure for XeI₂ involves considering several factors, including formal charges, octet rule exceptions, and the overall stability of the molecule. This comprehensive guide will delve into the process, evaluating different possible structures and justifying the selection of the most accurate representation.
Understanding Lewis Structures and the Octet Rule
A Lewis structure, also known as an electron dot structure, visually represents the bonding and lone pairs of electrons in a molecule. It helps predict the molecule's geometry and properties. The octet rule, a fundamental principle in chemistry, states that atoms tend to gain, lose, or share electrons to achieve a stable configuration of eight valence electrons. However, this rule has exceptions, especially with elements beyond the second period, such as xenon.
Possible Lewis Structures for XeI₂
Xenon has eight valence electrons, and each iodine atom has seven. Therefore, the total number of valence electrons in XeI₂ is 22 (8 + 7 + 7). Several Lewis structures can be drawn, but we must analyze each to determine the most stable and accurate representation.
Structure 1: Linear Structure with Expanded Octet on Xenon
This structure places the two iodine atoms linearly around the central xenon atom. Xenon is surrounded by two bonding pairs and three lone pairs, resulting in an expanded octet (10 electrons around xenon). This is depicted as:
I - Xe - I
Formal Charges: Xe (8-2-6=0), I (7-1-6=0). All formal charges are zero, which is favorable.
Structure 2: Bent Structure with Expanded Octet on Xenon
Another possibility is a bent structure, with the two iodine atoms positioned at an angle around the xenon atom. This also involves an expanded octet on xenon. A possible representation (though not the most stable):
I
|
Xe-I
Formal Charges: Similar to the linear structure, the formal charges would likely be zero for all atoms, depending on lone pair arrangement.
Structure 3: Structures Violating the Octet Rule (less likely)
While less likely, we could consider structures where the octet rule is violated for iodine, which is possible but less stable than structures with an expanded octet on Xenon. These structures are less favorable and will not be detailed here as the expanded octet for Xenon is a more likely occurrence.
Evaluating the Structures: Choosing the Best Lewis Structure
To choose the best Lewis structure, we need to consider several factors:
-
Formal Charges: Lower formal charges are generally preferred. A structure with formal charges of zero on all atoms is ideal. As both Structures 1 and 2 could achieve zero formal charges, this criterion alone cannot resolve the best option.
-
Octet Rule Exceptions: Elements in the third period and beyond can exceed the octet rule, and this is expected for Xenon in this case. Structures 1 and 2 accommodate this by expanding the valence shell of Xenon.
-
Electron Repulsion: VSEPR (Valence Shell Electron Pair Repulsion) theory suggests that electron pairs repel each other and will arrange themselves to minimize repulsion. A linear arrangement (Structure 1) minimizes repulsion compared to a bent arrangement (Structure 2).
-
Experimental Evidence: Experimental data, such as bond lengths and bond angles, strongly support a linear structure for XeI₂. This is crucial corroborating evidence for selection.
Why the Linear Structure (Structure 1) is the Best
Considering the factors above, the linear structure (Structure 1) emerges as the most plausible and accurate Lewis structure for XeI₂:
- Minimal Repulsion: The linear arrangement of the iodine atoms around xenon minimizes electron-electron repulsion.
- Zero Formal Charges: Both structures have zero formal charges.
- Expanded Octet: The expanded octet on xenon is fully accommodated, which is the most common observation for xenon compounds.
- Experimental Validation: Experimental evidence aligns with a linear molecular geometry for XeI₂, further supporting the choice of Structure 1.
Deeper Dive into VSEPR Theory and XeI₂ Geometry
VSEPR theory predicts the geometry of a molecule based on the number of electron pairs around the central atom. In XeI₂, xenon has five electron pairs: two bonding pairs and three lone pairs. According to VSEPR, this arrangement corresponds to a trigonal bipyramidal electron geometry.
However, the molecular geometry, which describes the arrangement of atoms only, is linear. The three lone pairs occupy the equatorial positions of the trigonal bipyramid, leaving the two iodine atoms at the axial positions, thus resulting in a linear molecular structure.
Applications and Further Considerations
Understanding the Lewis structure of XeI₂ is fundamental to grasping its chemical behavior and properties. This knowledge is crucial in various areas, including:
- Predicting reactivity: The Lewis structure helps predict how XeI₂ will react with other substances.
- Understanding bonding: The structure clarifies the nature of the Xe-I bonds (primarily covalent, influenced by the electronegativity difference).
- Spectroscopic analysis: The structure aids in interpreting spectroscopic data, such as infrared (IR) and Raman spectroscopy, to confirm the bonding and molecular geometry.
While XeI₂ is a relatively simple molecule, the process of determining its best Lewis structure highlights the importance of combining theoretical concepts (Lewis structures, VSEPR theory, formal charges) with experimental evidence for a complete and accurate representation.
Conclusion: The Importance of a Rigorous Approach
Determining the best Lewis structure for a molecule requires a multi-faceted approach. This detailed analysis of XeI₂ exemplifies the importance of considering formal charges, octet rule exceptions, electron pair repulsion, and available experimental data. The linear structure with an expanded octet on xenon (Structure 1) emerges as the most accurate and plausible representation of XeI₂, reflecting a sound understanding of chemical bonding principles. This meticulous process underscores the necessity of a rigorous and comprehensive approach to predicting and understanding molecular structures in chemistry. The understanding gained extends to the prediction of other xenon compounds properties and reactions. Further research in this area contributes to a broader understanding of the chemical behavior of noble gases.
Latest Posts
Latest Posts
-
A Companys Fiscal Year Must Correspond With The Calendar Year
Apr 03, 2025
-
In Recent Years The United States Has
Apr 03, 2025
-
Activity 9 5 Relief And Gradient Slope Analysis
Apr 03, 2025
-
Effectiveness And Efficiency Can Be Measured By
Apr 03, 2025
-
Which Of These Statements Is Not True
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Choose The Best Lewis Structure For Xei2 . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
