Which Of The Following Are Chemical Reactions
Holbox
Mar 31, 2025 · 6 min read
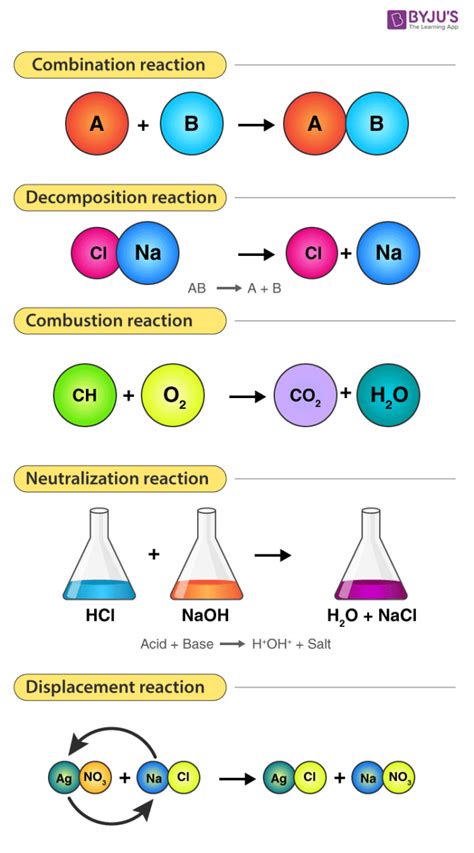
Table of Contents
- Which Of The Following Are Chemical Reactions
- Table of Contents
- Which of the Following Are Chemical Reactions? A Comprehensive Guide
- Understanding Chemical Reactions: The Fundamentals
- Key Indicators of Chemical Reactions:
- Differentiating Chemical Reactions from Physical Changes
- Analyzing Specific Examples: Are They Chemical Reactions?
- Advanced Considerations: Catalysis and Reaction Rates
- Conclusion: Mastering the Identification of Chemical Reactions
- Latest Posts
- Latest Posts
- Related Post
Which of the Following Are Chemical Reactions? A Comprehensive Guide
Identifying chemical reactions from a list of processes can seem daunting, but with a structured approach, it becomes straightforward. This comprehensive guide will delve into the core concepts of chemical reactions, providing you with the tools to confidently distinguish them from physical changes. We'll explore various examples and explain the key indicators that signify a chemical transformation. By the end, you'll be able to confidently analyze processes and determine if they represent chemical reactions.
Understanding Chemical Reactions: The Fundamentals
A chemical reaction is a process that leads to the transformation of one or more substances into one or more different substances. This transformation involves the rearrangement of atoms, breaking and forming chemical bonds. Crucially, the fundamental composition of the substances involved changes, resulting in new substances with different properties. This differs sharply from a physical change, where the substance's form or appearance alters, but its chemical composition remains the same.
Here's a breakdown of the key characteristics to look for when identifying a chemical reaction:
Key Indicators of Chemical Reactions:
-
Formation of a precipitate: A precipitate is a solid that forms from a solution during a chemical reaction. This often indicates a new substance has been created.
-
Evolution of a gas: The production of bubbles or a gas is a clear sign of a chemical change, signifying the formation of a gaseous product.
-
Change in temperature: Exothermic reactions release heat, causing a temperature increase, while endothermic reactions absorb heat, leading to a temperature decrease. A significant temperature change, either increase or decrease, is suggestive of a chemical reaction.
-
Change in color: A noticeable color change often indicates that a new substance with different properties has been formed.
-
Change in odor: The production of a new smell can signify a chemical reaction, as the formation of new compounds often results in unique odors.
-
Irreversibility (often, but not always): While not always definitive, many chemical reactions are difficult or impossible to reverse simply by changing physical conditions. This contrasts with many physical changes, which are easily reversed.
Differentiating Chemical Reactions from Physical Changes
The distinction between chemical and physical changes is crucial for understanding the nature of matter and its transformations. Let's examine some examples to illustrate this difference:
Physical Changes:
-
Melting ice: Ice (H₂O(s)) changes to liquid water (H₂O(l)). The chemical composition remains H₂O; only the state of matter changes.
-
Boiling water: Similar to melting ice, the chemical formula remains unchanged; only the state changes from liquid to gas.
-
Dissolving sugar in water: Sugar dissolves in water, forming a solution. However, the sugar molecules remain intact; they are simply dispersed throughout the water. Evaporation of the water would recover the original sugar.
-
Crushing a rock: The rock changes shape, but its chemical composition remains unaltered.
Chemical Reactions:
-
Burning wood: Wood combines with oxygen in the air, producing ash, gases (like carbon dioxide and water vapor), and heat. The chemical composition of the wood has fundamentally changed.
-
Rusting iron: Iron reacts with oxygen and water to form iron oxide (rust). This is a slow chemical reaction, but the iron's chemical composition changes irreversibly.
-
Baking a cake: The ingredients (flour, sugar, eggs, etc.) undergo complex chemical reactions, resulting in a completely new substance with different properties.
-
Digestion of food: Enzymes in the digestive system catalyze complex chemical reactions, breaking down food molecules into simpler substances that the body can absorb.
Analyzing Specific Examples: Are They Chemical Reactions?
Let's analyze several scenarios to determine whether they represent chemical reactions:
Scenario 1: Mixing baking soda and vinegar.
This is a classic example of a chemical reaction. Baking soda (sodium bicarbonate, NaHCO₃) reacts with acetic acid (CH₃COOH) in vinegar, producing carbon dioxide gas (CO₂), water (H₂O), and sodium acetate (CH₃COONa). The evolution of gas (CO₂), a clear change in temperature (often a slight cooling), and the formation of a new substance (sodium acetate) are all strong indicators of a chemical reaction.
Scenario 2: Dissolving salt in water.
This is a physical change. The salt (NaCl) dissolves in water, forming a solution. The salt molecules remain intact; they are simply separated and surrounded by water molecules. Evaporation of the water would recover the original salt. No new substance is formed.
Scenario 3: Burning a candle.
This is a chemical reaction. The wax (a complex hydrocarbon) reacts with oxygen in the air, producing carbon dioxide, water vapor, and heat. This combustion reaction involves a complete change in the chemical composition of the wax. The production of heat and light, as well as the formation of new gaseous substances, confirms this.
Scenario 4: Photosynthesis in plants.
This is a complex chemical reaction. Plants use sunlight, water, and carbon dioxide to produce glucose (a sugar) and oxygen. This is a vital process for life on Earth, and the production of a new substance (glucose) from different starting materials clearly indicates a chemical transformation.
Scenario 5: Mixing sand and water.
This is a physical change. Sand and water can be mixed, forming a suspension. However, neither substance undergoes a change in chemical composition. The sand particles are simply dispersed in the water.
Scenario 6: Electrolysis of water.
This is a chemical reaction. The application of an electric current to water breaks it down into its constituent elements: hydrogen gas (H₂) and oxygen gas (O₂). The production of distinct gases indicates the formation of new substances. This is a decomposition reaction.
Scenario 7: Melting chocolate.
This is a physical change. The chocolate changes from a solid to a liquid, but its chemical composition remains the same. Cooling the melted chocolate would restore its original solid form.
Scenario 8: Digestion of proteins.
This is a chemical reaction. Enzymes in the digestive system catalyze the hydrolysis of proteins, breaking down large protein molecules into smaller amino acids. This involves the breaking and reforming of chemical bonds, resulting in a change in chemical composition.
Scenario 9: Frying an egg.
This is a chemical reaction. The heat denatures the proteins in the egg white and yolk, causing them to change structure and solidify. This is an irreversible change in the chemical structure of the proteins.
Scenario 10: Mixing paint colors.
This is often a physical change, although some paints might undergo chemical reactions upon mixing. Typically, mixing paints results in a new color due to pigment dispersion and light absorption but doesn't involve a fundamental change in the chemical composition of the pigments themselves.
Advanced Considerations: Catalysis and Reaction Rates
Chemical reactions can be influenced by various factors, including catalysts and reaction conditions. A catalyst is a substance that speeds up a chemical reaction without being consumed in the process. Catalysts don't change the nature of the reaction, only its rate. Changes in temperature, pressure, and concentration can also affect the rate of chemical reactions but don't fundamentally alter whether a process is a chemical or physical change.
Conclusion: Mastering the Identification of Chemical Reactions
By understanding the key characteristics of chemical reactions and applying the examples provided, you can confidently identify chemical reactions from a given list of processes. Remember to look for evidence of the formation of new substances, significant temperature changes, gas evolution, color changes, odor changes, and irreversibility. Distinguishing between chemical reactions and physical changes is crucial for a solid foundation in chemistry and related fields. This guide serves as a stepping stone for further exploration into the fascinating world of chemical transformations.
Latest Posts
Latest Posts
-
A Firm Pursuing A Best Cost Provider Strategy
Apr 03, 2025
-
Below Are Incomplete Financial Statements For Bulldog Incorporated
Apr 03, 2025
-
Pn Pediatric Nursing Online Practice 2023 A
Apr 03, 2025
-
Find Req For The Circuit In Fig 2 94
Apr 03, 2025
-
Your Clients Target Demographic Is In Boston
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Which Of The Following Are Chemical Reactions . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
