What Is True About The Krebs Cycle
Holbox
Apr 01, 2025 · 6 min read
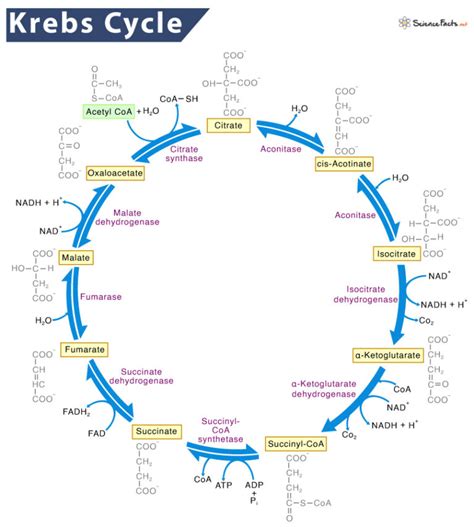
Table of Contents
- What Is True About The Krebs Cycle
- Table of Contents
- What's Really True About the Krebs Cycle: A Deep Dive into the Citric Acid Cycle
- The Core Function: Energy Generation and Precursor Synthesis
- Dispelling Myths: It's Not Just About ATP
- A Step-by-Step Look at the Reactions
- 1. Citrate Synthase: Condensation of Acetyl-CoA and Oxaloacetate
- 2. Aconitase: Isomerization of Citrate to Isocitrate
- 3. Isocitrate Dehydrogenase: Oxidative Decarboxylation of Isocitrate
- 4. α-Ketoglutarate Dehydrogenase: Oxidative Decarboxylation of α-Ketoglutarate
- 5. Succinyl-CoA Synthetase: Substrate-Level Phosphorylation
- 6. Succinate Dehydrogenase: Oxidation of Succinate
- 7. Fumarase: Hydration of Fumarate
- 8. Malate Dehydrogenase: Oxidation of Malate
- Regulation of the Krebs Cycle: A Delicate Balance
- Beyond Energy: The Krebs Cycle's Biosynthetic Roles
- Evolutionary Significance: A Universal Pathway
- Clinical Significance: Diseases and the Krebs Cycle
- Future Research: Unraveling the Nuances
- Conclusion: A Dynamic and Essential Pathway
- Latest Posts
- Latest Posts
- Related Post
What's Really True About the Krebs Cycle: A Deep Dive into the Citric Acid Cycle
The Krebs cycle, also known as the citric acid cycle (CAC) or the tricarboxylic acid cycle (TCA cycle), is a central metabolic pathway found in all aerobic organisms. It's a crucial link between glycolysis and oxidative phosphorylation, playing a vital role in energy production and providing building blocks for various biosynthetic pathways. While often simplified in introductory biology courses, the Krebs cycle is a complex and fascinating process with numerous intricacies and subtleties. This article delves deep into the realities of the Krebs cycle, dispelling common misconceptions and highlighting its multifaceted nature.
The Core Function: Energy Generation and Precursor Synthesis
The primary function of the Krebs cycle is undeniably energy generation. Through a series of eight enzyme-catalyzed reactions, it oxidizes acetyl-CoA, derived primarily from the breakdown of carbohydrates, fats, and proteins, to produce reduced electron carriers (NADH and FADH2) and a small amount of ATP (or GTP). These reduced carriers then feed into the electron transport chain, the powerhouse of cellular respiration, where the energy stored in their high-energy electrons is used to generate a significant amount of ATP via oxidative phosphorylation. This ATP fuels numerous cellular processes.
However, the Krebs cycle is much more than a simple energy factory. It acts as a critical metabolic hub, serving as a source of intermediates for various biosynthetic pathways. Many of the cycle's intermediates, such as oxaloacetate, α-ketoglutarate, and succinyl-CoA, are diverted to synthesize amino acids, nucleotides, fatty acids, and porphyrins (components of heme). This dual functionality underscores the Krebs cycle's importance in cellular metabolism.
Dispelling Myths: It's Not Just About ATP
A common misunderstanding is that the Krebs cycle's primary output is ATP. While it does generate a small amount of ATP (or GTP) directly through substrate-level phosphorylation, the vast majority of energy derived from the cycle is indirectly produced through the electron transport chain after the generation of NADH and FADH2. The direct ATP production is a minor component of the cycle’s overall contribution to energy metabolism.
A Step-by-Step Look at the Reactions
Let's examine each step of the Krebs cycle in detail, highlighting the key enzymes and reactions:
1. Citrate Synthase: Condensation of Acetyl-CoA and Oxaloacetate
The cycle begins with the condensation of acetyl-CoA (a two-carbon molecule) and oxaloacetate (a four-carbon molecule) to form citrate (a six-carbon molecule). This reaction, catalyzed by citrate synthase, is highly exergonic and irreversible, driving the entire cycle forward. The enzyme's active site undergoes a conformational change upon binding of oxaloacetate, facilitating the reaction.
2. Aconitase: Isomerization of Citrate to Isocitrate
Citrate, a tertiary alcohol, is then isomerized to isocitrate, a secondary alcohol, by aconitase. This involves a dehydration step followed by a rehydration step. Aconitase contains a [4Fe-4S] cluster, which is crucial for its catalytic activity and is sensitive to oxidative stress.
3. Isocitrate Dehydrogenase: Oxidative Decarboxylation of Isocitrate
Isocitrate dehydrogenase catalyzes the oxidative decarboxylation of isocitrate, producing α-ketoglutarate (a five-carbon molecule), CO2, and NADH. This is the first of two oxidative decarboxylation steps in the cycle and is a key regulatory point. The reaction requires Mn2+ or Mg2+ as a cofactor.
4. α-Ketoglutarate Dehydrogenase: Oxidative Decarboxylation of α-Ketoglutarate
Similar to the previous step, α-ketoglutarate dehydrogenase catalyzes the oxidative decarboxylation of α-ketoglutarate, yielding succinyl-CoA (a four-carbon molecule), CO2, and NADH. This complex enzyme shares structural and functional similarities with the pyruvate dehydrogenase complex.
5. Succinyl-CoA Synthetase: Substrate-Level Phosphorylation
Succinyl-CoA is converted to succinate by succinyl-CoA synthetase. This reaction involves substrate-level phosphorylation, producing GTP (in some organisms) or ATP. The energy released during thioester bond hydrolysis is used to phosphorylate GDP or ADP.
6. Succinate Dehydrogenase: Oxidation of Succinate
Succinate dehydrogenase catalyzes the oxidation of succinate to fumarate, producing FADH2. This enzyme is unique among the Krebs cycle enzymes because it's an integral part of the inner mitochondrial membrane and directly donates electrons to the electron transport chain.
7. Fumarase: Hydration of Fumarate
Fumarase catalyzes the hydration of fumarate to malate, adding a water molecule across the double bond. This reaction is stereospecific, producing only L-malate.
8. Malate Dehydrogenase: Oxidation of Malate
Finally, malate dehydrogenase oxidizes malate to oxaloacetate, producing NADH. This regenerates oxaloacetate, completing the cycle and allowing it to continue.
Regulation of the Krebs Cycle: A Delicate Balance
The Krebs cycle isn't a static process; its activity is tightly regulated to meet the cell's energy demands. Several factors influence its rate, including:
- Substrate Availability: The concentrations of acetyl-CoA and oxaloacetate directly affect the cycle's rate.
- Energy Charge: High levels of ATP and NADH inhibit several enzymes, slowing the cycle down, while ADP and NAD+ stimulate the cycle.
- Allosteric Regulation: Some enzymes, such as citrate synthase and isocitrate dehydrogenase, are subject to allosteric regulation by various metabolites.
- Covalent Modification: Some enzymes can be regulated through phosphorylation and dephosphorylation.
Beyond Energy: The Krebs Cycle's Biosynthetic Roles
The Krebs cycle's intermediates serve as precursors for numerous biosynthetic pathways:
- Amino Acid Synthesis: α-Ketoglutarate, oxaloacetate, and succinyl-CoA are essential precursors for the synthesis of various amino acids.
- Heme Synthesis: Succinyl-CoA is a crucial precursor in heme biosynthesis.
- Fatty Acid Synthesis: Citrate is transported out of the mitochondria and converted to acetyl-CoA for fatty acid synthesis.
- Gluconeogenesis: Oxaloacetate is an important precursor for gluconeogenesis, the synthesis of glucose from non-carbohydrate sources.
Evolutionary Significance: A Universal Pathway
The near-universality of the Krebs cycle across aerobic organisms highlights its fundamental role in cellular metabolism and its early evolutionary emergence. Slight variations exist in different organisms, reflecting adaptations to specific environmental conditions and metabolic needs. The cycle's efficiency and adaptability have made it a cornerstone of life as we know it.
Clinical Significance: Diseases and the Krebs Cycle
Dysfunction in the Krebs cycle can have significant clinical implications, often leading to various metabolic disorders. Mutations in enzymes involved in the cycle can result in energy deficiencies and accumulation of toxic intermediates. These deficiencies can manifest as various symptoms depending on the specific enzyme affected.
Future Research: Unraveling the Nuances
Despite decades of research, the Krebs cycle continues to be a subject of ongoing investigation. Researchers are continually uncovering new regulatory mechanisms, exploring the intricate interactions between the cycle and other metabolic pathways, and investigating the cycle's role in various diseases. Understanding the subtleties of this fundamental pathway is crucial for advancing our knowledge of cellular biology and developing new therapeutic strategies.
Conclusion: A Dynamic and Essential Pathway
The Krebs cycle is far more than a simple series of chemical reactions; it's a dynamic and intricate metabolic hub that plays a crucial role in energy production, biosynthesis, and cellular regulation. Its multifaceted nature and importance in various biological processes continue to fascinate researchers and underscore its fundamental role in life. This comprehensive overview highlights the complexity and significance of the Krebs cycle, dispelling common misconceptions and emphasizing its crucial role in maintaining cellular homeostasis and enabling life's diverse processes. Further research will undoubtedly continue to reveal new aspects of this vital metabolic pathway.
Latest Posts
Latest Posts
-
Select The True Statements About The Citric Acid Cycle
Apr 04, 2025
-
An Example Of A Period Cost Is
Apr 04, 2025
-
Are Fetal Pig Toes Split Or Fused
Apr 04, 2025
-
Chain Ganglia Are Part Of The
Apr 04, 2025
-
Identify A Lateral Projection Of A Vertebra
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about What Is True About The Krebs Cycle . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
