Select The Correct Proper Or Common Name For The Compound
Holbox
Mar 31, 2025 · 5 min read
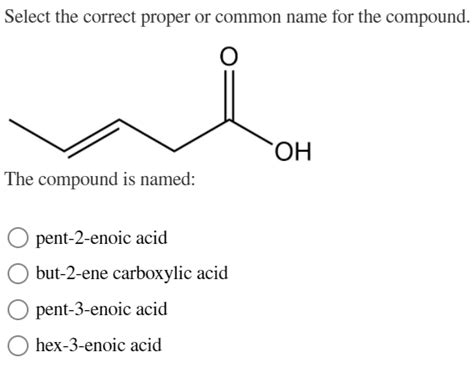
Table of Contents
- Select The Correct Proper Or Common Name For The Compound
- Table of Contents
- Selecting the Correct Proper or Common Name for a Compound: A Comprehensive Guide
- Understanding Chemical Nomenclature: IUPAC vs. Common Names
- Navigating the IUPAC Nomenclature System
- 1. Inorganic Compounds:
- 2. Organic Compounds:
- Common Names and Their Contextual Use
- 1. Everyday Life:
- 2. Specific Industries:
- 3. Historical Context:
- Potential Pitfalls of Using Common Names
- When to Use IUPAC vs. Common Names
- Conclusion: A Balanced Approach
- Latest Posts
- Latest Posts
- Related Post
Selecting the Correct Proper or Common Name for a Compound: A Comprehensive Guide
Naming chemical compounds might seem like a straightforward task, but it's a surprisingly complex field with intricate rules and exceptions. The correct nomenclature is crucial for clear communication within the scientific community, avoiding ambiguity and ensuring everyone is talking about the same substance. This comprehensive guide delves into the nuances of selecting the appropriate proper or common name for a chemical compound, covering various naming systems and providing practical examples.
Understanding Chemical Nomenclature: IUPAC vs. Common Names
The primary system for naming chemical compounds is the International Union of Pure and Applied Chemistry (IUPAC) nomenclature. IUPAC provides a standardized set of rules that allows for the unambiguous naming of any chemical compound, regardless of its complexity. These names are often systematic, meaning they directly reflect the compound's structure and composition. For example, the IUPAC name for table salt is sodium chloride, clearly indicating its constituent elements.
However, alongside IUPAC nomenclature, many compounds also possess common names. These names are often shorter, more convenient, and historically established, sometimes predating the systematic IUPAC system. While common names are widely used and understood within specific contexts, they can be ambiguous and even lead to confusion if used interchangeably across different disciplines or regions. For instance, ethanol is the IUPAC name for ethyl alcohol, a common name many are familiar with.
The choice between using a proper (IUPAC) or common name depends heavily on the context. Scientific publications strongly favor IUPAC names to ensure clarity and precision. In contrast, everyday settings, informal discussions, or specific industries might prefer common names for simplicity. Understanding this distinction is crucial to avoid miscommunication and ensure the accurate identification of compounds.
Navigating the IUPAC Nomenclature System
The IUPAC system offers several approaches to naming compounds, depending on their chemical nature:
1. Inorganic Compounds:
Inorganic compounds generally follow simpler rules than organic compounds. They are typically named based on their constituent elements and their oxidation states. For example:
-
Binary compounds: These consist of two elements. The less electronegative element is named first, followed by the more electronegative element with an "-ide" suffix. Examples include:
- NaCl (sodium chloride)
- MgO (magnesium oxide)
- H₂S (hydrogen sulfide)
-
Ternary compounds: These consist of three elements. The naming conventions become more complex, often involving Roman numerals to specify the oxidation state of the central metal ion. Examples include:
- FeCl₃ (iron(III) chloride)
- CuSO₄ (copper(II) sulfate)
- KMnO₄ (potassium permanganate)
-
Acids and Bases: Acids are generally named based on the anion they form. For instance, HCl (hydrochloric acid) forms the chloride ion. Bases often have hydroxide (OH⁻) as a constituent.
2. Organic Compounds:
Organic compounds are far more diverse and complex than inorganic ones, requiring a more sophisticated naming system. IUPAC nomenclature for organic compounds considers:
-
Parent chain: The longest continuous carbon chain in the molecule forms the basis of the name.
-
Substituents: Any branches or functional groups attached to the parent chain are named as substituents.
-
Numbering: The carbon atoms in the parent chain are numbered to indicate the position of substituents, ensuring the lowest possible numbers are used.
-
Functional groups: The presence of specific functional groups (like alcohols, ketones, carboxylic acids, etc.) dictates suffixes and prefixes in the name.
Examples include:
- CH₃CH₂CH₃: propane (simple alkane)
- CH₃CH₂OH: ethanol (alcohol)
- CH₃COOH: ethanoic acid (carboxylic acid)
- CH₃COCH₃: propan-2-one (ketone)
Common Names and Their Contextual Use
While IUPAC names provide unambiguous identification, common names are frequently used due to their familiarity and brevity. It's crucial to understand when using common names is acceptable and when it's necessary to revert to IUPAC nomenclature. Here are some common scenarios:
1. Everyday Life:
In everyday settings, common names dominate. Few people refer to sodium chloride as "sodium chloride" in their kitchen; instead, they use "table salt." Similarly, "baking soda" (sodium bicarbonate) and "vinegar" (acetic acid) are common names widely understood by the general public.
2. Specific Industries:
Certain industries have established conventions for using common names. The pharmaceutical industry, for instance, often uses common names or brand names for drugs that are otherwise known by their IUPAC names. Similarly, the food industry might use common names for additives or ingredients.
3. Historical Context:
Some compounds have historical common names that are so deeply ingrained that they persist even in scientific literature. For example, benzene is rarely referred to by its systematic IUPAC name (cyclohexa-1,3,5-triene).
Potential Pitfalls of Using Common Names
While common names offer convenience, using them without caution can lead to several problems:
-
Ambiguity: A single common name might refer to multiple compounds, leading to confusion.
-
Regional variations: Common names can vary regionally, causing miscommunication across different geographical areas.
-
Lack of structural information: Unlike IUPAC names, common names do not inherently provide information about the compound's structure.
-
Inconsistent usage: The use of common names can be inconsistent across different fields and publications.
When to Use IUPAC vs. Common Names
The decision of whether to use IUPAC or common names depends significantly on the target audience and the context:
-
Scientific publications and research: IUPAC nomenclature is the gold standard. Clarity and precision are paramount in scientific communication, leaving no room for ambiguity.
-
Educational settings: Both IUPAC and common names might be used, depending on the level and the specific subject matter. Introducing both allows for a comprehensive understanding of chemical nomenclature.
-
General public and non-scientific contexts: Common names are often preferred for simplicity and ease of understanding. However, it's important to ensure the common name is widely understood and unambiguous in the context.
-
Industry-specific applications: Industry conventions and established practices might dictate the use of either IUPAC or common names.
Conclusion: A Balanced Approach
Selecting the correct proper or common name for a chemical compound requires a nuanced understanding of both IUPAC nomenclature and common naming conventions. While IUPAC names provide the precision and clarity necessary for scientific communication, common names offer convenience and familiarity in everyday life and specific industries. A balanced approach, combining the strengths of both systems, ensures effective communication and avoids the pitfalls associated with ambiguous or inconsistent naming practices. By understanding the context and the target audience, you can confidently choose the most appropriate name for any chemical compound. Always prioritize clarity and avoid ambiguity to ensure effective communication within the scientific and wider community.
Latest Posts
Latest Posts
-
What Is The Equivalent Resistance Between Points A And B
Apr 03, 2025
-
A Customer Is Traveling To A Branch Office
Apr 03, 2025
-
Setting Up The Math For A Two Step Quantitative Problem
Apr 03, 2025
-
What Key Role Does An Investment Bank Play
Apr 03, 2025
-
Which Bacterial Strain Is The Least Competitively Dominant
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Select The Correct Proper Or Common Name For The Compound . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
