Peptidoglycan Is A Unique Macromolecule Found In Bacterial
Holbox
Mar 30, 2025 · 7 min read
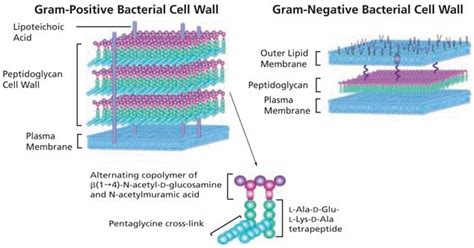
Table of Contents
- Peptidoglycan Is A Unique Macromolecule Found In Bacterial
- Table of Contents
- Peptidoglycan: The Unique Macromolecule Defining Bacteria
- The Molecular Architecture of Peptidoglycan: A Rigid Meshwork
- 1. Glycan Chains: The Sugar Backbone
- 2. Peptide Cross-Links: Strengthening the Structure
- The Significance of Peptidoglycan in Bacterial Cell Biology
- 1. Maintaining Cell Shape and Integrity:
- 2. Protecting against Osmotic Lysis:
- 3. Facilitating Cell Growth and Division:
- 4. Interaction with other cell wall components:
- Peptidoglycan Biosynthesis: A Target for Antibiotics
- Variations in Peptidoglycan Structure and its Implications
- Gram-positive vs. Gram-negative Bacteria:
- Peptidoglycan and the Immune System: A Complex Interaction
- Conclusion: Peptidoglycan – A Keystone in Bacterial Biology and Antibiotic Development
- Latest Posts
- Latest Posts
- Related Post
Peptidoglycan: The Unique Macromolecule Defining Bacteria
Peptidoglycan, also known as murein, is a unique macromolecule found exclusively in the cell walls of bacteria. This crucial component plays a pivotal role in maintaining bacterial cell shape, providing structural integrity, and protecting the cell from osmotic lysis. Understanding peptidoglycan's structure, biosynthesis, and function is fundamental to comprehending bacterial biology and developing effective antibacterial strategies. This comprehensive article delves deep into the world of peptidoglycan, exploring its intricate composition, its significance in bacterial survival, and its implications for medical research and advancements in antibiotic development.
The Molecular Architecture of Peptidoglycan: A Rigid Meshwork
Peptidoglycan's remarkable strength and rigidity stem from its unique architecture. It's a cross-linked polymer composed of two main components:
1. Glycan Chains: The Sugar Backbone
The glycan strands form the backbone of peptidoglycan. These chains are composed of alternating units of N-acetylglucosamine (NAG) and N-acetylmuramic acid (NAM), linked together by β-1,4 glycosidic bonds. This repeating disaccharide unit is crucial for the structural stability of the entire molecule. The precise arrangement and linkage of NAG and NAM are essential for the overall integrity and functionality of the peptidoglycan layer.
2. Peptide Cross-Links: Strengthening the Structure
The strength of peptidoglycan isn't solely reliant on the glycan chains. Short peptide chains are attached to the NAM units, creating a three-dimensional network. The composition and length of these peptide chains vary across different bacterial species, contributing to the diversity observed in bacterial cell walls. These peptides are often composed of four to five amino acids, commonly including D-amino acids, which are rarely found in proteins of eukaryotic organisms. This unique feature has significant implications for the targeting of peptidoglycan biosynthesis by antibiotics.
The peptide chains are cross-linked to each other, creating a robust mesh-like structure. This cross-linking is mediated by transpeptidases, also known as penicillin-binding proteins (PBPs), which catalyze the formation of peptide bonds between adjacent peptide chains. This intricate cross-linking process is crucial for the overall structural integrity and rigidity of the peptidoglycan layer. The degree of cross-linking can vary depending on the bacterial species and environmental conditions, influencing the overall strength and flexibility of the cell wall.
The Significance of Peptidoglycan in Bacterial Cell Biology
Peptidoglycan's function extends beyond simply providing structural support. It plays a crucial role in several essential bacterial processes:
1. Maintaining Cell Shape and Integrity:
The rigid peptidoglycan layer provides the essential structural support that dictates the characteristic shape of bacterial cells. Whether a bacterium is a coccus (spherical), bacillus (rod-shaped), or spirillum (spiral-shaped), the peptidoglycan layer is instrumental in defining and maintaining this shape. This is especially crucial in resisting the turgor pressure exerted by the internal contents of the cell. Without the peptidoglycan layer, the bacterial cell would lyse under osmotic stress.
2. Protecting against Osmotic Lysis:
Bacteria often inhabit environments with varying osmotic pressures. The peptidoglycan layer acts as a protective barrier against osmotic stress, preventing the cell from bursting due to the influx of water. This is particularly important in hypotonic environments where the water potential outside the cell is higher than inside the cell, leading to a net influx of water. The rigidity of the peptidoglycan layer prevents excessive expansion and subsequent lysis of the bacterial cell.
3. Facilitating Cell Growth and Division:
During cell growth and division, the peptidoglycan layer must be remodeled and expanded. Specific enzymes are involved in this process, including autolysins, which break down existing peptidoglycan bonds, and transglycosylases and transpeptidases, which synthesize new peptidoglycan. This coordinated process ensures that the peptidoglycan layer expands evenly and maintains its structural integrity during bacterial growth and division. A carefully regulated balance between synthesis and degradation is crucial for proper bacterial cell growth.
4. Interaction with other cell wall components:
In Gram-positive bacteria, the peptidoglycan layer is significantly thicker and is associated with other cell wall components, such as teichoic acids and lipoteichoic acids. These components play important roles in cell wall stability, ion binding, and interactions with the host immune system. In Gram-negative bacteria, the peptidoglycan layer is thinner and is located in the periplasmic space, between the inner and outer membranes. The outer membrane, composed of lipopolysaccharide (LPS), adds another layer of protection and plays an important role in bacterial virulence.
Peptidoglycan Biosynthesis: A Target for Antibiotics
The biosynthesis of peptidoglycan is a complex, multi-step process that involves several enzymes. This process has become a major target for antibiotics, as inhibiting its crucial steps leads to bacterial cell death. The key enzymes involved are:
- Transglycosylases: These enzymes catalyze the formation of glycosidic bonds between NAG and NAM, extending the glycan chains.
- Transpeptidases (PBPs): These enzymes, as mentioned earlier, catalyze the cross-linking of peptide chains, creating the strong, rigid meshwork of peptidoglycan.
- Bactoprenol: This lipid carrier transports peptidoglycan precursors across the cytoplasmic membrane to the site of peptidoglycan synthesis.
Many antibiotics target these enzymes, effectively disrupting peptidoglycan synthesis and leading to bacterial cell lysis. Beta-lactam antibiotics, including penicillin and cephalosporins, are well-known examples. These antibiotics competitively inhibit transpeptidases, preventing the cross-linking of peptide chains and weakening the peptidoglycan structure. Glycopeptides, such as vancomycin, also inhibit peptidoglycan synthesis by binding to the D-Ala-D-Ala terminus of peptidoglycan precursors, preventing their incorporation into the growing peptidoglycan layer.
Variations in Peptidoglycan Structure and its Implications
While the basic structure of peptidoglycan is conserved across bacterial species, there are variations in the composition and cross-linking of the peptide chains. These variations contribute to the diversity observed in bacterial cell walls and can affect the susceptibility of bacteria to different antibiotics.
Gram-positive vs. Gram-negative Bacteria:
A significant distinction lies in the thickness of the peptidoglycan layer. Gram-positive bacteria possess a thick peptidoglycan layer, often accounting for up to 90% of their cell wall mass. This thick layer is essential for their resistance to osmotic stress and also contributes to their overall robustness. In contrast, Gram-negative bacteria have a thin peptidoglycan layer located in the periplasmic space between the inner and outer membranes. The outer membrane of Gram-negative bacteria presents an additional barrier to antibiotics, contributing to their greater resistance to certain antimicrobial agents.
Peptidoglycan and the Immune System: A Complex Interaction
Peptidoglycan plays a crucial role in the interaction between bacteria and the host immune system. Fragments of peptidoglycan, known as peptidoglycan fragments or muropeptides, are released during bacterial growth and death. These fragments are potent immunostimulants, activating various components of the innate and adaptive immune systems.
They can trigger the production of pro-inflammatory cytokines, leading to the recruitment of immune cells to the site of infection. Moreover, they can activate the complement system, contributing to bacterial killing. Recognition of peptidoglycan by immune cells involves various pattern recognition receptors, including peptidoglycan recognition proteins (PGRPs) and NOD-like receptors (NLRs). Understanding the interaction between peptidoglycan and the immune system is essential for developing new strategies to combat bacterial infections.
Conclusion: Peptidoglycan – A Keystone in Bacterial Biology and Antibiotic Development
Peptidoglycan, a unique macromolecule found in all bacteria, is essential for maintaining bacterial cell shape, integrity, and survival. Its intricate structure, robust cross-linking, and crucial role in bacterial growth have made it a primary target for antibiotic development. Variations in peptidoglycan structure across different bacterial species contribute to the diverse responses observed to antibiotics and highlight the complexity of bacterial cell wall biology. Continued research into peptidoglycan biosynthesis, structure, and interaction with the immune system will pave the way for the development of novel antibacterial strategies to combat bacterial infections and address the growing problem of antibiotic resistance. The continued study of this fascinating molecule is essential for advancing our understanding of bacterial pathogenesis and for developing effective treatments for bacterial infections. Further investigation into the diverse structures and functions of peptidoglycan across various bacterial species promises to reveal further insights into the evolution and adaptation of these crucial microorganisms. The unique properties of peptidoglycan make it a critical target for research and development in the fight against bacterial infections, emphasizing its importance in both basic biology and clinical applications.
Latest Posts
Latest Posts
-
Which Of The Following Is An Example Of Operant Conditioning
Apr 03, 2025
-
Olfaction And Hearing Are Processed In The
Apr 03, 2025
-
Economists Have Difficulty Applying The Scientific Method Because
Apr 03, 2025
-
Criminal Justice In America 10th Edition
Apr 03, 2025
-
A Companys Strategy Is At Full Power
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Peptidoglycan Is A Unique Macromolecule Found In Bacterial . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
