Label The Parts Of The Immunoglobulin
Holbox
Apr 01, 2025 · 6 min read
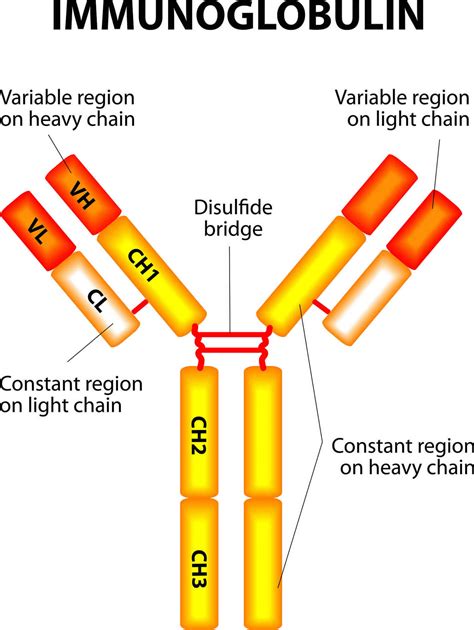
Table of Contents
- Label The Parts Of The Immunoglobulin
- Table of Contents
- Label the Parts of the Immunoglobulin: A Deep Dive into Antibody Structure and Function
- The Basic Structure: A Y-Shaped Marvel
- 1. Heavy Chains (H Chains): The Backbone of the Antibody
- 2. Light Chains (L Chains): Adding Specificity to the Binding Site
- Functional Regions and Their Roles
- 1. Antigen-Binding Site (Paratope): The Key to Recognition
- 2. Hinge Region: Flexibility and Functionality
- 3. Fc Region (Fragment Crystallizable): Effector Function Powerhouse
- 4. Fab Region (Fragment Antigen-Binding): The Antigen-Grabbing Arms
- Immunoglobulin Isotypes: A Functional Diversity
- Beyond the Basics: Antibody Engineering and Therapeutic Applications
- Conclusion: A Complex Molecule with a Vital Role
- Latest Posts
- Latest Posts
- Related Post
Label the Parts of the Immunoglobulin: A Deep Dive into Antibody Structure and Function
Immunoglobulins (Ig), also known as antibodies, are glycoprotein molecules produced by plasma cells (activated B cells) that play a crucial role in the adaptive immune system. Understanding their structure is key to understanding their function. This article will provide a comprehensive guide to labeling the various parts of an immunoglobulin, delving into their specific roles and significance in immune responses.
The Basic Structure: A Y-Shaped Marvel
Immunoglobulins are characterized by their distinctive Y-shape, comprising four polypeptide chains: two identical heavy chains (H chains) and two identical light chains (L chains). These chains are held together by strong disulfide bonds, creating a robust and functional molecule.
1. Heavy Chains (H Chains): The Backbone of the Antibody
The heavy chains form the backbone of the immunoglobulin, dictating its isotype (IgM, IgG, IgA, IgD, IgE) and determining its effector functions. Each heavy chain contains:
-
Variable Region (VH): Located at the N-terminal end of the heavy chain, the variable region is highly diverse, enabling antibodies to recognize a vast array of antigens. This region is responsible for antigen binding. Within the VH region, three hypervariable regions, also known as complementarity-determining regions (CDRs), are particularly important for antigen recognition. These CDRs form the antigen-binding site or paratope. The precise arrangement of amino acids within the CDRs determines the specificity of the antibody for a particular antigen. Mutations in these regions can drastically alter antibody affinity and specificity.
-
Constant Region (CH): The constant region of the heavy chain determines the antibody isotype and effector functions. Different isotypes have different constant regions, contributing to their diverse roles in the immune system. For example, IgG antibodies are involved in opsonization and complement activation, while IgE antibodies mediate allergic reactions. The constant region interacts with various immune cells and proteins, triggering downstream effects. The CH region is further subdivided into CH1, CH2, CH3 (and CH4 in some isotypes). The hinge region lies between CH1 and CH2, offering flexibility to the antibody structure.
2. Light Chains (L Chains): Adding Specificity to the Binding Site
The light chains contribute to the antigen-binding site, adding to the specificity of the antibody. There are two types of light chains:
- Kappa (κ) Chains: These are more prevalent in humans.
- Lambda (λ) Chains: These are also found in humans, though generally less abundant than kappa chains.
Each light chain contains:
-
Variable Region (VL): Similar to the VH region, the VL region is highly variable and plays a crucial role in antigen binding. It contains three CDRs that contribute to the formation of the antigen-binding site.
-
Constant Region (CL): The constant region of the light chain does not exhibit the same isotype variation as the heavy chain. It primarily contributes to the overall structure and stability of the antibody molecule.
Functional Regions and Their Roles
The overall structure of the immunoglobulin translates directly into its diverse functions. Understanding these functions requires appreciating the specific roles of different regions:
1. Antigen-Binding Site (Paratope): The Key to Recognition
The antigen-binding site is formed by the variable regions of both the heavy and light chains (VH and VL). The precise three-dimensional arrangement of amino acid residues within this site determines the antibody's affinity and specificity for a particular antigen. This incredibly specific interaction is crucial for neutralizing pathogens and triggering other immune responses. The strength of the interaction (affinity) dictates how effectively the antibody can bind and neutralize its target.
2. Hinge Region: Flexibility and Functionality
The hinge region, located between the CH1 and CH2 domains of the heavy chain, provides flexibility to the immunoglobulin molecule. This flexibility allows the antibody to bind to antigens with different orientations and shapes. The hinge region also contains cysteine residues that form disulfide bonds, stabilizing the antibody structure. Proteolytic cleavage in this region can generate smaller antibody fragments with distinct functions.
3. Fc Region (Fragment Crystallizable): Effector Function Powerhouse
The Fc region consists of the constant regions of the heavy chains (CH2 and CH3, and potentially CH4). This region doesn't directly participate in antigen binding but plays a vital role in mediating effector functions. The Fc region interacts with various immune cells and proteins, leading to:
-
Complement Activation: Binding of the Fc region to complement proteins initiates the complement cascade, a powerful system that leads to pathogen lysis and enhanced phagocytosis.
-
Opsonization: The Fc region can bind to Fc receptors (FcRs) on phagocytic cells such as macrophages and neutrophils, enhancing their ability to recognize and engulf pathogens coated with antibodies.
-
Antibody-Dependent Cell-mediated Cytotoxicity (ADCC): The Fc region can bind to FcRs on natural killer (NK) cells and other cytotoxic cells, leading to the destruction of antibody-coated target cells.
4. Fab Region (Fragment Antigen-Binding): The Antigen-Grabbing Arms
The Fab region comprises the variable and constant regions of both the heavy and light chains (VH, VL, CH1, and CL). It forms the two "arms" of the Y-shaped antibody and is responsible for antigen recognition and binding. Cleavage of the antibody molecule by papain yields two Fab fragments and one Fc fragment.
Immunoglobulin Isotypes: A Functional Diversity
The five main immunoglobulin isotypes (IgM, IgG, IgA, IgD, IgE) exhibit distinct structural and functional characteristics. The differences primarily reside in the constant region of the heavy chain:
-
IgM: The first antibody produced during an immune response. It is a pentamer (five antibody units joined together), possessing a high avidity (overall binding strength) for antigen. It is a potent activator of the complement system.
-
IgG: The most abundant antibody isotype in the blood. It provides long-lasting immunity and plays roles in opsonization, complement activation, and ADCC. There are four subclasses of IgG (IgG1, IgG2, IgG3, IgG4), each with subtle differences in effector functions.
-
IgA: The predominant antibody in mucosal secretions (saliva, tears, breast milk). It protects mucosal surfaces from pathogens. It exists as a monomer, dimer, or trimer.
-
IgD: Its function is not fully understood, but it is found on the surface of B cells and may play a role in B cell activation.
-
IgE: Mediates allergic reactions and defense against parasites. It binds to mast cells and basophils, triggering the release of histamine and other inflammatory mediators.
Beyond the Basics: Antibody Engineering and Therapeutic Applications
The detailed understanding of immunoglobulin structure has paved the way for significant advancements in antibody engineering. Scientists can now modify antibodies to enhance their affinity, specificity, and effector functions, resulting in the development of highly effective therapeutic antibodies used to treat a wide range of diseases, including cancer, autoimmune disorders, and infectious diseases. These engineered antibodies often have improved half-lives or are designed to target specific cell types or pathways.
Conclusion: A Complex Molecule with a Vital Role
Immunoglobulins are complex glycoproteins with a highly organized structure that directly dictates their function. By understanding the distinct roles of the heavy and light chains, the variable and constant regions, the hinge region, and the Fc and Fab fragments, we gain a deeper appreciation of how these molecules contribute to a robust and effective immune system. This knowledge is crucial not only for fundamental immunology research but also for the development of novel therapeutic strategies utilizing antibodies. The ongoing research into immunoglobulin structure and function continues to reveal new insights and possibilities for treating various diseases.
Latest Posts
Latest Posts
-
What Makes The Sunglasses Option Appealing
Apr 04, 2025
-
The Conflict Perspective Views Social Inequality As
Apr 04, 2025
-
Product Repositioning Means Changing The Products Position
Apr 04, 2025
-
On November 10 Of The Current Year Flores
Apr 04, 2025
-
Acceleration Due To Gravity On Venus
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Label The Parts Of The Immunoglobulin . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
