Draw The Product S Of The Following Reaction
Holbox
Apr 02, 2025 · 5 min read
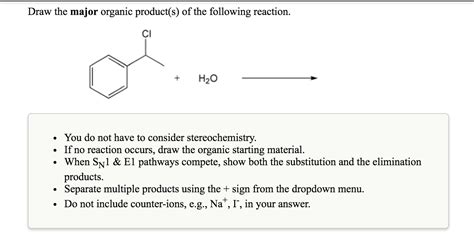
Table of Contents
- Draw The Product S Of The Following Reaction
- Table of Contents
- Predicting Reaction Products: A Comprehensive Guide to Organic Chemistry
- A. Functional Groups: The Heart of Reactivity
- B. Reaction Mechanisms: The "How" of Reactions
- C. Reaction Conditions: The "Where" and "When"
- A. Example 1: Addition of HBr to an Alkene
- B. Example 2: Oxidation of a Secondary Alcohol
- C. Example 3: SN1 and SN2 Reactions
- D. Example 4: Elimination Reactions
- E. Example 5: Esterification
- F. Example 6: Grignard Reaction
- Latest Posts
- Latest Posts
- Related Post
Predicting Reaction Products: A Comprehensive Guide to Organic Chemistry
Predicting the products of a chemical reaction is a cornerstone of organic chemistry. It requires a deep understanding of functional groups, reaction mechanisms, and the interplay of various factors influencing reactivity. This article delves into the process of predicting reaction products, covering key concepts and providing a structured approach to tackle diverse scenarios. We'll explore several reaction types, highlighting crucial aspects like regioselectivity and stereoselectivity. While we won't be able to cover every possible reaction, the principles discussed here are broadly applicable.
I. Understanding the Fundamentals
Before diving into specific reactions, let's establish a strong foundation:
A. Functional Groups: The Heart of Reactivity
Functional groups are specific atoms or groups of atoms within a molecule that are responsible for its characteristic chemical reactions. Recognizing functional groups is the first crucial step in predicting reaction products. Common functional groups include:
- Alcohols (-OH): Undergo reactions like oxidation, dehydration, and esterification.
- Aldehydes (-CHO) and Ketones (-C=O): Can be reduced, undergo nucleophilic addition, and form various derivatives.
- Carboxylic Acids (-COOH): React to form esters, amides, and anhydrides.
- Amines (-NH2, -NHR, -NR2): Act as bases and undergo reactions like alkylation and acylation.
- Alkenes (C=C) and Alkynes (C≡C): Participate in addition reactions (e.g., hydrogenation, halogenation, hydrohalogenation).
- Haloalkanes (R-X, where X is a halogen): Undergo nucleophilic substitution and elimination reactions.
- Ethers (R-O-R'): Relatively unreactive compared to other functional groups, but can be cleaved under specific conditions.
B. Reaction Mechanisms: The "How" of Reactions
Understanding the reaction mechanism – the step-by-step process by which reactants transform into products – is crucial for accurate product prediction. Mechanisms often involve:
- Nucleophilic attack: A nucleophile (electron-rich species) attacks an electrophile (electron-deficient species).
- Electrophilic attack: An electrophile attacks a nucleophile.
- Addition reactions: Atoms are added across a multiple bond (e.g., alkenes, alkynes).
- Substitution reactions: One atom or group is replaced by another.
- Elimination reactions: A small molecule (e.g., water, HCl) is removed from the reactant, often forming a multiple bond.
- Rearrangements: Atoms within a molecule shift positions, leading to structural changes.
C. Reaction Conditions: The "Where" and "When"
Reaction conditions significantly influence product formation. Key factors include:
- Temperature: Higher temperatures often favor faster reactions and sometimes lead to different products (e.g., kinetic vs. thermodynamic control).
- Solvent: The solvent can affect reaction rates and selectivity. Polar solvents favor polar reactions, while non-polar solvents favor non-polar reactions.
- Catalyst: Catalysts accelerate reactions without being consumed themselves, often influencing selectivity.
- Reagent stoichiometry: The relative amounts of reactants can dictate which products are formed.
II. Predicting Products: A Step-by-Step Approach
Let's now apply these principles to predicting the products of various reactions. To illustrate, we'll use hypothetical examples, but the underlying principles are universally applicable. Remember to always consider the functional groups present, the likely mechanism, and the reaction conditions.
A. Example 1: Addition of HBr to an Alkene
Reaction: 1-butene + HBr → ?
Analysis: This is an electrophilic addition reaction. HBr adds across the double bond. Markovnikov's rule dictates that the hydrogen atom adds to the carbon atom with more hydrogen atoms already attached, while the bromine adds to the other carbon.
Product: 2-bromobutane
B. Example 2: Oxidation of a Secondary Alcohol
Reaction: 2-pentanol + [O] → ?
Analysis: Secondary alcohols are oxidized to ketones. The oxidizing agent ([O]) removes two hydrogen atoms from the alcohol group, forming a carbonyl group.
Product: 2-pentanone
C. Example 3: SN1 and SN2 Reactions
Reaction 1 (SN1): tert-butyl bromide + NaOH (in aqueous ethanol) → ?
Analysis: tert-butyl bromide is a tertiary halide, favoring the SN1 mechanism (unimolecular nucleophilic substitution). A carbocation intermediate is formed, which is then attacked by the hydroxide ion.
Product: tert-butyl alcohol
Reaction 2 (SN2): methyl bromide + NaOH (in acetone) → ?
Analysis: Methyl bromide is a primary halide, favoring the SN2 mechanism (bimolecular nucleophilic substitution). The hydroxide ion attacks the carbon atom bearing the bromine, resulting in a direct displacement.
Product: methanol
D. Example 4: Elimination Reactions
Reaction: 2-bromobutane + KOH (alcoholic) → ?
Analysis: This reaction is an elimination reaction, specifically E2 (bimolecular elimination). The strong base (KOH) abstracts a proton from a carbon adjacent to the carbon bearing the bromine, leading to the formation of a double bond and the expulsion of HBr. Zaitsev's rule predicts the formation of the more substituted alkene.
Product: 2-butene (major product), 1-butene (minor product)
E. Example 5: Esterification
Reaction: Ethanoic acid + ethanol (acid catalyst) → ?
Analysis: This is an esterification reaction. The carboxylic acid reacts with the alcohol to form an ester and water. The acid catalyst (often sulfuric acid) facilitates the reaction.
Product: Ethyl ethanoate + water
F. Example 6: Grignard Reaction
Reaction: Bromobenzene + Methylmagnesium bromide (Grignard reagent) → ? followed by acid workup
Analysis: Grignard reagents are powerful nucleophiles. The Grignard reagent adds to the carbonyl group of the aldehyde or ketone. In this case, the reaction would need a carbonyl-containing compound. Let's assume we're reacting it with benzaldehyde. The Grignard reagent attacks the carbonyl carbon, followed by an acid workup to protonate the alkoxide.
Product (assuming reaction with benzaldehyde): 1,2-diphenylethanol
III. Advanced Considerations
Predicting reaction products can become significantly more complex when dealing with:
- Multiple Functional Groups: The presence of multiple functional groups can lead to competing reactions or unexpected outcomes. Determining the relative reactivity of different functional groups is essential.
- Stereochemistry: Reactions can lead to the formation of stereoisomers (enantiomers or diastereomers). Understanding stereoselectivity (preferential formation of one stereoisomer over another) is crucial.
- Reaction Kinetics vs. Thermodynamics: Kinetic control favors the faster reaction, while thermodynamic control favors the more stable product. Temperature and reaction time can influence the outcome.
- Complex Reaction Mechanisms: Some reactions involve intricate multi-step mechanisms with intermediate species. A thorough understanding of these mechanisms is needed for accurate prediction.
IV. Conclusion
Predicting the products of chemical reactions is a multifaceted skill that combines knowledge of functional groups, reaction mechanisms, and reaction conditions. The ability to accurately predict products is fundamental to designing and executing organic syntheses. While the examples provided here illustrate some common reaction types, continuous learning and practice are crucial for mastering this essential aspect of organic chemistry. By systematically applying the principles outlined in this article, you will significantly improve your ability to accurately predict and understand the products of a wide range of chemical reactions. Remember to always consult reliable textbooks and resources for detailed information and specific reaction details. The practice problems and examples available in organic chemistry textbooks are invaluable tools for improving your predictive skills. Good luck!
Latest Posts
Latest Posts
-
The Velocity Components Of An Incompressible Two Dimensional
Apr 05, 2025
-
An Event That Could Cause Harm Or Loss Is A
Apr 05, 2025
-
Identify The True And False Statements About Race
Apr 05, 2025
-
Which Question Can Be Answered Using Business Intelligence
Apr 05, 2025
-
One Of Your Customers Is Delinquent
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Draw The Product S Of The Following Reaction . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
