Draw The Product Of The Following Reaction
Holbox
Apr 01, 2025 · 6 min read
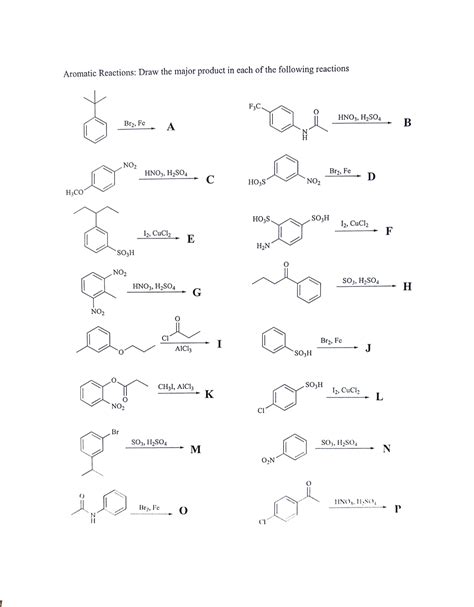
Table of Contents
- Draw The Product Of The Following Reaction
- Table of Contents
- Predicting Organic Reaction Products: A Comprehensive Guide
- Understanding Reaction Mechanisms: The Key to Prediction
- Common Reaction Types and Predictive Strategies
- Factors Affecting Reaction Outcome
- Advanced Techniques and Considerations
- Conclusion
- Latest Posts
- Latest Posts
- Related Post
Predicting Organic Reaction Products: A Comprehensive Guide
Predicting the outcome of organic reactions is a cornerstone of organic chemistry. This skill, honed through understanding reaction mechanisms and functional group transformations, is crucial for designing syntheses and interpreting experimental results. This article will delve into the process of predicting products, focusing on different reaction types and the factors that influence their outcome. We'll explore several examples, providing a detailed analysis of each reaction and its predicted product(s). Remember, without a specific reaction provided, I cannot draw a specific product. Instead, I will provide a framework for tackling such problems, encompassing various common reaction types.
Understanding Reaction Mechanisms: The Key to Prediction
Before predicting the product of any reaction, a deep understanding of its mechanism is paramount. The mechanism outlines the step-by-step process of bond breaking and bond formation, revealing the pathway by which reactants are transformed into products. Key concepts to grasp include:
- Nucleophiles and Electrophiles: Nucleophiles, electron-rich species, attack electron-deficient electrophiles. Understanding the relative nucleophilicity and electrophilicity of reactants is essential.
- Leaving Groups: Leaving groups are atoms or groups that readily depart from a molecule, often taking a pair of electrons with them. Good leaving groups are generally weak bases (e.g., halides, tosylates).
- Stereochemistry: Reactions can lead to the formation of stereoisomers (enantiomers or diastereomers). Understanding stereochemistry is crucial for predicting the configuration of the product.
- Reaction Conditions: Reaction conditions (temperature, solvent, catalyst, etc.) significantly influence the reaction pathway and the product(s) formed.
Common Reaction Types and Predictive Strategies
Let's examine some frequently encountered reaction types and strategies for predicting their products:
1. SN1 and SN2 Reactions: These nucleophilic substitution reactions involve the replacement of a leaving group by a nucleophile.
-
SN2 Reactions: These are concerted reactions where the nucleophile attacks the carbon atom bearing the leaving group from the backside, leading to inversion of configuration. Steric hindrance significantly affects the rate of SN2 reactions. Strong nucleophiles favor SN2 reactions.
-
SN1 Reactions: These are two-step reactions involving the formation of a carbocation intermediate. The rate of SN1 reactions is dependent on the stability of the carbocation. Weak nucleophiles and tertiary alkyl halides favor SN1 reactions. Racemization (formation of a mixture of enantiomers) is often observed in SN1 reactions.
Predictive Strategy: Identify the substrate (alkyl halide, alcohol, etc.), nucleophile, and leaving group. Consider steric factors and the strength of the nucleophile. Determine whether SN1 or SN2 conditions are favored based on the substrate structure and reaction conditions. Predict the product considering stereochemistry.
Example: The reaction of 2-bromobutane with sodium hydroxide (NaOH) in ethanol is likely to proceed via SN2, yielding 2-butanol with inverted configuration.
2. E1 and E2 Reactions: These elimination reactions involve the removal of a leaving group and a proton from adjacent carbon atoms, leading to the formation of a double bond (alkene).
-
E2 Reactions: These are concerted reactions where the base abstracts a proton and the leaving group departs simultaneously. Strong bases favor E2 reactions. Stereochemistry plays a significant role, with anti-periplanar geometry preferred.
-
E1 Reactions: These are two-step reactions involving the formation of a carbocation intermediate followed by proton abstraction. Weak bases and tertiary alkyl halides favor E1 reactions. Zaitsev's rule often predicts the major product (the more substituted alkene).
Predictive Strategy: Identify the substrate, base, and leaving group. Consider the strength of the base and the substrate structure. Determine whether E1 or E2 conditions are favored. Predict the product(s) considering Zaitsev's rule and stereochemistry.
Example: The reaction of 2-bromo-2-methylpropane with potassium tert-butoxide (t-BuOK) in tert-butanol will likely proceed via E2, yielding 2-methylpropene as the major product (Zaitsev's rule).
3. Addition Reactions: These reactions involve the addition of atoms or groups to a double or triple bond.
-
Electrophilic Addition: Electrophiles add to the double bond, forming a carbocation intermediate, which is then attacked by a nucleophile. Markovnikov's rule predicts the regioselectivity (where the electrophile adds).
-
Nucleophilic Addition: Nucleophiles add to the carbonyl group (C=O) of aldehydes and ketones.
Predictive Strategy: Identify the reactant with the double or triple bond and the reagents being added. Consider Markovnikov's rule for electrophilic additions and the reactivity of the carbonyl group for nucleophilic additions.
Example: The addition of hydrogen bromide (HBr) to propene will follow Markovnikov's rule, yielding 2-bromopropane.
4. Oxidation and Reduction Reactions: These reactions involve the change in oxidation state of a carbon atom.
-
Oxidation: Increases the oxidation state of carbon (e.g., primary alcohol to aldehyde to carboxylic acid). Oxidizing agents include potassium permanganate (KMnO4), chromic acid (H2CrO4), and Jones reagent.
-
Reduction: Decreases the oxidation state of carbon (e.g., ketone to alcohol). Reducing agents include lithium aluminum hydride (LiAlH4) and sodium borohydride (NaBH4).
Predictive Strategy: Identify the functional group being oxidized or reduced and the oxidizing or reducing agent used. Determine the resulting change in oxidation state.
Example: The oxidation of ethanol with potassium dichromate (K2Cr2O7) in acidic medium will yield acetic acid.
5. Grignard Reactions: Grignard reagents (RMgX) are powerful nucleophiles that react with carbonyl compounds (aldehydes, ketones, esters, and carbon dioxide) to form new carbon-carbon bonds.
Predictive Strategy: Identify the Grignard reagent and the carbonyl compound. Predict the addition of the Grignard reagent to the carbonyl group, followed by acid workup to yield the alcohol.
Example: The reaction of methylmagnesium bromide (CH3MgBr) with formaldehyde (HCHO) followed by acid workup will yield ethanol.
Factors Affecting Reaction Outcome
Several factors can influence the outcome of a reaction, including:
- Solvent: Polar protic solvents favor SN1 and E1 reactions, while polar aprotic solvents favor SN2 reactions.
- Temperature: Higher temperatures often favor elimination reactions over substitution reactions.
- Steric Hindrance: Bulky groups can hinder the approach of nucleophiles or bases, affecting reaction rates and regioselectivity.
- Catalyst: Catalysts can accelerate reactions and alter reaction pathways.
Advanced Techniques and Considerations
For more complex reactions, advanced techniques are needed:
- Retrosynthetic Analysis: Working backward from the desired product to identify suitable starting materials and reactions.
- Spectroscopic Techniques: NMR, IR, and mass spectrometry can be used to confirm the structure of the product.
Conclusion
Predicting the products of organic reactions requires a solid understanding of reaction mechanisms, functional group transformations, and the various factors influencing reaction outcomes. By systematically analyzing the reactants, reaction conditions, and potential pathways, one can develop the skills to accurately predict the products formed. Remember, practice is key. The more reactions you analyze, the better you'll become at predicting the outcomes. This comprehensive framework should empower you to approach various reaction types with confidence and accurately predict their products. Always remember to consider all possible reaction pathways and the factors that might influence the outcome.
Latest Posts
Latest Posts
-
Lab Aids 603s Investigating And Applying Genetics Answer Key
Apr 05, 2025
-
Ned Owns A Small Electronics Repair Business
Apr 05, 2025
-
Given The Single Step Reaction Shown Draw The Curved Arrow Mechanism
Apr 05, 2025
-
Which Statement About The Field Of Abnormal Psychology Is Accurate
Apr 05, 2025
-
Cpg Islands Are Associated With Which Of The Following
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Draw The Product Of The Following Reaction . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
