Draw The Organic Product Of The Reaction
Holbox
Apr 05, 2025 · 6 min read
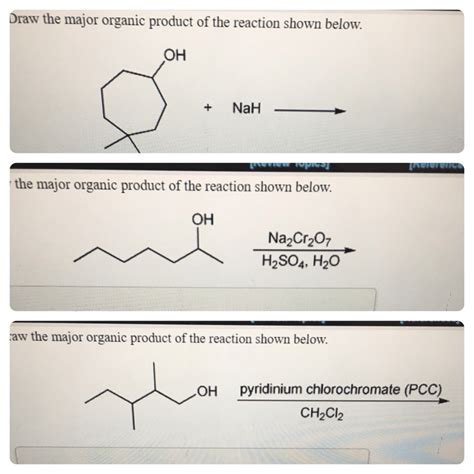
Table of Contents
- Draw The Organic Product Of The Reaction
- Table of Contents
- Drawing the Organic Product of a Reaction: A Comprehensive Guide
- Understanding Reaction Mechanisms
- 1. Nucleophilic Attack and Electrophilic Attack
- 2. Electron Flow and Curly Arrows
- 3. Intermediates and Transition States
- 4. Stereochemistry
- Common Reaction Types and Product Prediction
- 1. Acid-Base Reactions
- 2. Nucleophilic Substitution Reactions (SN1 and SN2)
- 3. Elimination Reactions (E1 and E2)
- 4. Addition Reactions
- 5. Oxidation and Reduction Reactions
- 6. Grignard Reactions
- 7. Aldol Condensation
- Strategies for Drawing Organic Products
- Advanced Topics and Considerations
- Latest Posts
- Latest Posts
- Related Post
Drawing the Organic Product of a Reaction: A Comprehensive Guide
Predicting the organic product of a reaction is a fundamental skill in organic chemistry. It requires a thorough understanding of reaction mechanisms, functional group transformations, and stereochemistry. This comprehensive guide will walk you through the process, providing strategies and examples to help you master this crucial skill. We'll cover a range of reactions, from simple acid-base reactions to more complex multi-step processes.
Understanding Reaction Mechanisms
Before attempting to predict the product, it's crucial to understand the underlying reaction mechanism. The mechanism dictates the pathway by which reactants transform into products. Key concepts include:
1. Nucleophilic Attack and Electrophilic Attack
Many organic reactions involve nucleophiles (electron-rich species) attacking electrophiles (electron-deficient species). Understanding which atom in a molecule is nucleophilic and which is electrophilic is key to predicting the outcome. For example, in a Grignard reaction, the carbon atom in the Grignard reagent acts as a nucleophile, attacking the electrophilic carbonyl carbon of a ketone or aldehyde.
2. Electron Flow and Curly Arrows
Using curly arrows to depict the movement of electrons is crucial for visualizing reaction mechanisms. These arrows show the flow of electrons from a nucleophile to an electrophile or from a bond to form a new bond. Accurately drawing curly arrows will help you predict the product and avoid common mistakes.
3. Intermediates and Transition States
Many reactions proceed through intermediate species (e.g., carbocations, carbanions) that are not stable enough to be isolated but play a crucial role in the reaction pathway. Understanding the stability of these intermediates can help you predict the most likely product. Transition states represent the highest energy point in the reaction pathway.
4. Stereochemistry
Stereochemistry is essential for accurately drawing the product. Consider whether the reaction proceeds with retention, inversion, or racemization of stereochemistry. This is particularly important for reactions involving chiral centers.
Common Reaction Types and Product Prediction
Let's explore some common organic reactions and strategies for predicting their products:
1. Acid-Base Reactions
Acid-base reactions are among the simplest organic reactions. They involve the transfer of a proton (H⁺) from an acid to a base. The product is the conjugate acid of the base and the conjugate base of the acid.
Example: The reaction between acetic acid (CH₃COOH) and sodium hydroxide (NaOH):
CH₃COOH + NaOH → CH₃COO⁻Na⁺ + H₂O
The product is sodium acetate and water.
2. Nucleophilic Substitution Reactions (SN1 and SN2)
These reactions involve the substitution of a leaving group by a nucleophile. SN1 reactions proceed through a carbocation intermediate, leading to racemization at the chiral center (if present). SN2 reactions proceed through a concerted mechanism with inversion of configuration at the chiral center.
Example (SN2): The reaction between bromomethane (CH₃Br) and sodium hydroxide (NaOH):
CH₃Br + NaOH → CH₃OH + NaBr
The product is methanol and sodium bromide. The reaction proceeds with inversion of configuration if the carbon atom is chiral.
Example (SN1): Tertiary butyl bromide reacting with water:
(CH₃)₃CBr + H₂O → (CH₃)₃COH + HBr
The product is tert-butanol and HBr. A carbocation intermediate is formed, leading to racemization if the starting material was chiral.
3. Elimination Reactions (E1 and E2)
Elimination reactions involve the removal of a leaving group and a proton from adjacent carbon atoms, leading to the formation of a double bond (alkene). E1 reactions proceed through a carbocation intermediate, while E2 reactions are concerted. Zaitsev's rule predicts the major product in elimination reactions: the more substituted alkene is generally favored.
Example (E2): The reaction between 2-bromopropane and potassium hydroxide:
CH₃CHBrCH₃ + KOH → CH₃CH=CH₂ + KBr + H₂O
The major product is propene.
4. Addition Reactions
Addition reactions involve the addition of atoms or groups to a double or triple bond. Markovnikov's rule predicts the regioselectivity of addition reactions to unsymmetrical alkenes: the more substituted carbon atom gets the electrophile.
Example: The addition of hydrogen bromide (HBr) to propene:
CH₃CH=CH₂ + HBr → CH₃CHBrCH₃
The major product is 2-bromopropane.
5. Oxidation and Reduction Reactions
Oxidation involves the loss of electrons or an increase in oxidation state, while reduction involves the gain of electrons or a decrease in oxidation state. Many oxidizing and reducing agents are used in organic chemistry, and their reactivity varies.
Example (Oxidation): The oxidation of a primary alcohol to a carboxylic acid using strong oxidizing agents like potassium permanganate (KMnO₄) or chromic acid (H₂CrO₄).
Example (Reduction): The reduction of a ketone to a secondary alcohol using reducing agents like sodium borohydride (NaBH₄) or lithium aluminum hydride (LiAlH₄).
6. Grignard Reactions
Grignard reactions involve the reaction of a Grignard reagent (RMgX) with a carbonyl compound (aldehyde, ketone, ester, or acid chloride). The product depends on the type of carbonyl compound.
Example: The reaction of methylmagnesium bromide (CH₃MgBr) with formaldehyde (HCHO):
CH₃MgBr + HCHO → CH₃CH₂OMgBr → CH₃CH₂OH (after acidic workup)
The product is ethanol.
7. Aldol Condensation
Aldol condensation involves the reaction of two carbonyl compounds (aldehydes or ketones) to form a β-hydroxy carbonyl compound, which can then dehydrate to form an α,β-unsaturated carbonyl compound.
Example: The aldol condensation of acetaldehyde:
2 CH₃CHO → CH₃CH(OH)CH₂CHO → CH₃CH=CHCHO + H₂O
The product is crotonaldehyde.
Strategies for Drawing Organic Products
To effectively draw the organic product of a reaction, follow these steps:
- Identify the functional groups: Determine the functional groups present in the reactants.
- Identify the type of reaction: Classify the reaction (e.g., SN1, SN2, addition, elimination).
- Determine the mechanism: Understand the stepwise process of the reaction.
- Predict the intermediate(s): If applicable, predict the structure of any intermediates formed.
- Predict the product: Draw the structure of the final product, considering stereochemistry if relevant.
- Check for resonance stabilization: If applicable, consider if the product can be stabilized through resonance.
- Consider side reactions: Be aware of possible side reactions that could lead to the formation of byproducts.
Advanced Topics and Considerations
- Protecting Groups: Sometimes, functional groups need protection to prevent unwanted reactions. Understanding protecting group strategies is crucial for complex syntheses.
- Regioselectivity and Stereoselectivity: Many reactions exhibit regioselectivity (preference for one regioisomer over another) and stereoselectivity (preference for one stereoisomer over another).
- Multistep Syntheses: Planning multistep syntheses requires careful consideration of reaction compatibility and yield.
Mastering the art of predicting organic products is a gradual process that requires consistent practice and a thorough understanding of reaction mechanisms. By following these strategies and continually practicing with diverse examples, you'll develop the skills necessary to confidently predict the outcome of various organic reactions. Remember to always double-check your work and consider all possibilities. The more you practice, the better you will become!
Latest Posts
Latest Posts
-
One Advantage Of City States In Mesopotamia Was That
Apr 10, 2025
-
Investment X Offers To Pay You 4700
Apr 10, 2025
-
If A Typical Aspirin Tablet Contains 325
Apr 10, 2025
-
Nitrifying Bacteria Convert To
Apr 10, 2025
-
Spatial Vis Slopes And Curves Answers
Apr 10, 2025
Related Post
Thank you for visiting our website which covers about Draw The Organic Product Of The Reaction . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.