Draw The Major Product Of This Reaction
Holbox
Mar 31, 2025 · 5 min read
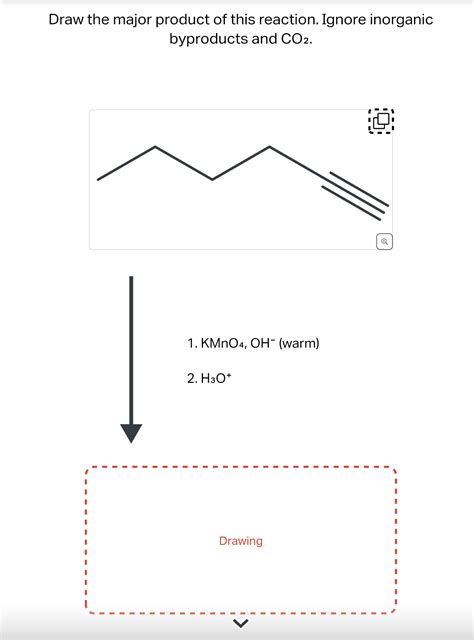
Table of Contents
- Draw The Major Product Of This Reaction
- Table of Contents
- Draw the Major Product of This Reaction: A Comprehensive Guide
- Understanding Reaction Mechanisms: The Key to Predicting Products
- Factors Influencing Product Selectivity
- Examples: Predicting Major Products in Various Reactions
- 1. SN1 and SN2 Reactions
- 2. E1 and E2 Elimination Reactions
- 3. Addition Reactions
- 4. Electrophilic Aromatic Substitution
- 5. Grignard Reactions
- Advanced Considerations
- Conclusion
- Latest Posts
- Latest Posts
- Related Post
Draw the Major Product of This Reaction: A Comprehensive Guide
Predicting the major product of a chemical reaction is a fundamental skill in organic chemistry. It requires a deep understanding of reaction mechanisms, functional group reactivity, and the principles of thermodynamics and kinetics. This article will delve into the process of determining the major product, exploring various reaction types and the factors influencing product selectivity. We'll examine several examples, emphasizing the reasoning behind each prediction.
Understanding Reaction Mechanisms: The Key to Predicting Products
Before we jump into specific examples, it's crucial to grasp the concept of reaction mechanisms. A reaction mechanism outlines the step-by-step process of bond breaking and bond formation during a chemical transformation. Understanding the mechanism allows us to predict the likely intermediates and ultimately, the major product. Different mechanisms lead to different products, even with the same starting materials and reagents. Key concepts include:
- Nucleophilic attack: A nucleophile (electron-rich species) attacks an electrophile (electron-deficient species).
- Electrophilic attack: An electrophile attacks a nucleophile.
- Elimination reactions: Loss of a leaving group and a proton, often resulting in the formation of a double bond.
- Addition reactions: Addition of atoms or groups to a multiple bond (e.g., double or triple bond).
- Rearrangements: Changes in the carbon skeleton of a molecule, often driven by carbocation stability.
Factors Influencing Product Selectivity
Several factors determine which product is formed predominantly in a reaction:
- Thermodynamics: The most stable product is often favored, reflecting the principle of minimizing Gibbs free energy. This is especially relevant in reactions at equilibrium.
- Kinetics: The reaction pathway with the lowest activation energy is usually faster, even if it doesn't lead to the most thermodynamically stable product. Kinetic control often dominates at lower temperatures.
- Steric hindrance: Bulky groups can hinder the approach of reagents, affecting the reaction rate and product distribution.
- Solvent effects: The solvent can influence the stability of intermediates and transition states, impacting product selectivity.
- Reagent control: The choice of reagent can significantly influence the reaction pathway and the resulting product.
Examples: Predicting Major Products in Various Reactions
Let's examine several reaction types and illustrate the process of predicting the major product.
1. SN1 and SN2 Reactions
These reactions are common nucleophilic substitutions.
-
SN1 (Substitution Nucleophilic Unimolecular): A two-step mechanism involving carbocation formation. The rate depends only on the concentration of the substrate. More substituted carbocations are more stable, leading to greater selectivity for more substituted products. Racemization is often observed due to the planar nature of the carbocation intermediate.
-
SN2 (Substitution Nucleophilic Bimolecular): A one-step concerted mechanism. The rate depends on the concentration of both the substrate and the nucleophile. Steric hindrance plays a crucial role; SN2 reactions are favored with less hindered substrates. Inversion of configuration is typically observed.
Example: Consider the reaction of 2-bromobutane with sodium hydroxide (NaOH) in ethanol.
-
SN1: In a polar protic solvent like ethanol, a carbocation intermediate forms. The nucleophile (OH⁻) can attack from either side, leading to a racemic mixture of 2-butanol.
-
SN2: With a strong nucleophile like NaOH, an SN2 reaction is more likely, leading to inversion of configuration at the chiral carbon, resulting in (S)-2-butanol if the starting material was (R)-2-bromobutane. However, the strength of the nucleophile and the steric hindrance around the carbon determine whether SN1 or SN2 will be favored.
2. E1 and E2 Elimination Reactions
These reactions involve the removal of a leaving group and a proton to form a double bond (alkene).
-
E1 (Elimination Unimolecular): A two-step mechanism involving carbocation formation. The rate depends only on the concentration of the substrate. More substituted alkenes (Zaitsev's rule) are usually favored due to greater stability.
-
E2 (Elimination Bimolecular): A one-step concerted mechanism. The rate depends on the concentration of both the substrate and the base. Stereochemistry is important; the leaving group and the proton must be anti-periplanar for efficient E2 elimination.
Example: Dehydration of 2-methyl-2-propanol with concentrated sulfuric acid.
This reaction proceeds via an E1 mechanism due to the tertiary carbocation formed. The most substituted alkene, 2-methylpropene, is the major product, following Zaitsev's rule.
3. Addition Reactions
These reactions involve the addition of atoms or groups to a multiple bond. Markovnikov's rule often applies to electrophilic additions to alkenes.
Example: Addition of HBr to propene.
The proton adds to the less substituted carbon (Markovnikov's rule), leading to 2-bromopropane as the major product. The reason is the formation of a more stable secondary carbocation intermediate.
4. Electrophilic Aromatic Substitution
These reactions involve the substitution of a hydrogen atom on an aromatic ring by an electrophile. The directing effects of substituents are crucial in determining the position of substitution.
Example: Nitration of toluene.
The methyl group is an ortho/para-directing activator. Therefore, the major products are ortho-nitrotoluene and para-nitrotoluene, with para-nitrotoluene often being the major isomer due to less steric hindrance.
5. Grignard Reactions
These reactions involve the addition of a Grignard reagent (organomagnesium halide) to a carbonyl group.
Example: Reaction of bromobenzene Grignard reagent with formaldehyde.
The Grignard reagent acts as a nucleophile, attacking the carbonyl carbon of formaldehyde. After acidic workup, the major product is benzyl alcohol.
Advanced Considerations
- Regioselectivity: The preference for the formation of one constitutional isomer over another.
- Stereoselectivity: The preference for the formation of one stereoisomer over another (e.g., enantioselectivity or diastereoselectivity).
- Chemoselectivity: The preferential reaction of one functional group over another in a molecule containing multiple functional groups.
Conclusion
Predicting the major product of a chemical reaction is a multifaceted process requiring a thorough understanding of reaction mechanisms, thermodynamics, kinetics, and steric effects. By carefully considering these factors and applying the relevant rules (like Markovnikov's rule and Zaitsev's rule), we can accurately predict the outcome of many organic reactions. Practice is key to mastering this crucial skill. Remember to always analyze the reactants, reagents, reaction conditions, and the likely mechanism to arrive at a well-justified prediction. This comprehensive approach, combined with a deep understanding of organic chemistry principles, will significantly improve your ability to correctly predict the major product of any given reaction. Continuous study and problem-solving are essential for developing expertise in this area.
Latest Posts
Latest Posts
-
For The Query Boston Italian Food
Apr 03, 2025
-
After Malaria Is Cured The Frequency Of The Hbs Allele
Apr 03, 2025
-
Why Should Marketers Be Aware Of The Bric Countries
Apr 03, 2025
-
Google Draw Is Its Cloud First Approach
Apr 03, 2025
-
If Your Check Of The Person Leads You To Suspect
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Draw The Major Product Of This Reaction . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
