Columns On The Periodic Table Are Called
Holbox
Apr 01, 2025 · 7 min read
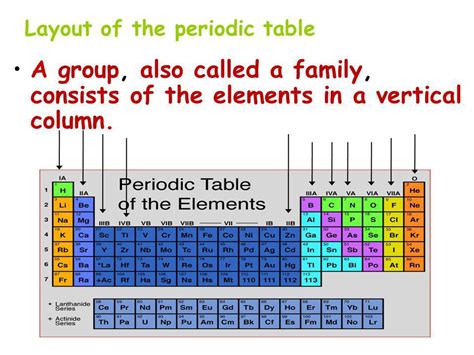
Table of Contents
- Columns On The Periodic Table Are Called
- Table of Contents
- Columns on the Periodic Table are Called Groups (or Families): A Deep Dive into Chemical Properties
- Understanding the Periodic Table's Structure: Rows vs. Columns
- The Significance of Group Number: Valence Electrons and Chemical Behavior
- Exploring the Main Groups: A Closer Look at Chemical Properties
- Group 1: Alkali Metals
- Group 2: Alkaline Earth Metals
- Group 17: Halogens
- Group 18: Noble Gases
- Transition Metals: A Complex Group with Varied Properties
- Inner Transition Metals (Lanthanides and Actinides): Rare Earth Elements
- Trends Across Periods and Groups: Predicting Chemical Behavior
- The Importance of Groups in Chemistry and Beyond
- Conclusion: Groups – The Key to Understanding Chemical Behavior
- Latest Posts
- Latest Posts
- Related Post
Columns on the Periodic Table are Called Groups (or Families): A Deep Dive into Chemical Properties
The periodic table, a cornerstone of chemistry, organizes elements based on their atomic structure and properties. Understanding its organization is crucial for comprehending chemical behavior and predicting reactions. While many focus on the rows (periods), the columns, known as groups or families, are equally vital because they highlight elements sharing similar chemical properties. This article delves into the fascinating world of groups on the periodic table, exploring their characteristics, trends, and significance in various applications.
Understanding the Periodic Table's Structure: Rows vs. Columns
Before we dive into the specifics of groups, let's briefly review the periodic table's structure. The table arranges elements in a grid, with each element occupying a unique square. The rows, or periods, represent increasing energy levels of electrons. As you move across a period from left to right, the number of electrons in the outermost shell (valence electrons) increases, influencing the element's reactivity.
The columns, or groups, are the focus of our discussion. Elements within the same group possess the same number of valence electrons. This shared electron configuration dictates their similar chemical behavior, explaining why elements within a group often exhibit comparable reactivity and form similar types of compounds.
The Significance of Group Number: Valence Electrons and Chemical Behavior
The group number directly relates to the number of valence electrons an element possesses. This is especially true for the main group elements (Groups 1-2 and 13-18). For example:
- Group 1 (Alkali Metals): Elements in this group have one valence electron, making them highly reactive. They readily lose this electron to form +1 ions.
- Group 2 (Alkaline Earth Metals): These elements possess two valence electrons, leading to a slightly lower reactivity compared to alkali metals. They typically form +2 ions.
- Group 17 (Halogens): With seven valence electrons, halogens are highly reactive nonmetals. They readily gain one electron to form -1 ions, achieving a stable octet.
- Group 18 (Noble Gases): These elements have a full valence shell (eight electrons, except for helium with two), making them exceptionally unreactive. Their stability is a direct result of their complete electron configuration.
Understanding this relationship between group number and valence electrons is fundamental to predicting how elements will interact and the types of compounds they'll form.
Exploring the Main Groups: A Closer Look at Chemical Properties
Let's explore some key groups in more detail, highlighting their characteristic properties and notable applications:
Group 1: Alkali Metals
Alkali metals are soft, silvery-white, highly reactive metals. Their reactivity stems from their single valence electron, readily lost to form +1 ions. This leads to vigorous reactions with water and air. Key characteristics include:
- Low density: They are lighter than water.
- Low melting and boiling points: They are relatively easy to melt.
- Good conductors of heat and electricity: This makes them useful in various electrical applications.
Applications: Sodium (Na) and potassium (K) are essential for biological processes. Lithium (Li) is used in batteries due to its high electrochemical potential.
Group 2: Alkaline Earth Metals
Slightly less reactive than alkali metals, alkaline earth metals have two valence electrons, forming +2 ions. They are harder and denser than alkali metals and have higher melting and boiling points. Key characteristics include:
- Relatively high melting and boiling points: Compared to alkali metals.
- Good conductors of heat and electricity: Similar to alkali metals.
- Form stable compounds: They readily react with other elements.
Applications: Magnesium (Mg) is used in lightweight alloys. Calcium (Ca) is crucial for bone structure and various biological processes.
Group 17: Halogens
Halogens are highly reactive nonmetals with seven valence electrons, readily gaining one electron to form -1 ions. This high reactivity makes them excellent oxidizing agents. Key characteristics include:
- Highly reactive: They readily react with metals and other nonmetals.
- Varied physical states: Fluorine (F) and chlorine (Cl) are gases at room temperature, bromine (Br) is a liquid, and iodine (I) is a solid.
- Form diatomic molecules: They exist as pairs of atoms (e.g., Cl₂).
Applications: Chlorine (Cl) is used in water purification. Iodine (I) is essential for thyroid function. Fluorine (F) is used in dental products.
Group 18: Noble Gases
Noble gases are exceptionally unreactive due to their complete valence shells. This inertness makes them ideal for various applications where non-reactivity is essential. Key characteristics include:
- Inertness: They rarely participate in chemical reactions.
- Colorless, odorless, and tasteless gases: They are generally unremarkable in their physical properties.
- Low boiling points: They exist as gases at room temperature.
Applications: Helium (He) is used in balloons and MRI machines. Argon (Ar) is used in welding and as a protective atmosphere. Neon (Ne) is used in lighting.
Transition Metals: A Complex Group with Varied Properties
Transition metals occupy the central block of the periodic table (Groups 3-12). Unlike main group elements, transition metals exhibit more complex chemical behavior due to the involvement of d-electrons in bonding. Key characteristics include:
- Variable oxidation states: They can form ions with different charges.
- Formation of colored compounds: Many of their compounds are brightly colored.
- Catalytic activity: Many transition metals and their compounds are excellent catalysts.
- High melting and boiling points: Generally higher than main group metals.
Applications: Iron (Fe) is crucial for steel production. Copper (Cu) is used in electrical wiring. Platinum (Pt) and palladium (Pd) are used as catalysts in various industrial processes.
Inner Transition Metals (Lanthanides and Actinides): Rare Earth Elements
Located at the bottom of the periodic table, the lanthanides and actinides are collectively known as inner transition metals. They have unique electronic configurations with filling of f-orbitals. Key characteristics include:
- Similar chemical properties: Elements within each series exhibit similar chemical behavior.
- Radioactive nature (Actinides): Most actinides are radioactive.
- Use in specialized applications: Their unique properties make them valuable in specific technologies.
Applications: Lanthanides are used in various alloys and magnets. Certain actinides are used in nuclear reactors and medical applications (though with caution due to their radioactivity).
Trends Across Periods and Groups: Predicting Chemical Behavior
Understanding the trends in properties across periods and groups allows chemists to predict the behavior of elements. These trends are influenced by factors such as atomic radius, electronegativity, and ionization energy.
- Atomic Radius: Generally decreases across a period (left to right) and increases down a group (top to bottom).
- Electronegativity: Generally increases across a period and decreases down a group.
- Ionization Energy: Generally increases across a period and decreases down a group.
These trends are crucial for predicting reactivity and the types of bonds elements will form.
The Importance of Groups in Chemistry and Beyond
The organization of the periodic table into groups is not just a convenient arrangement; it's a fundamental tool for understanding the behavior of matter. The similarities in properties within a group allow chemists to predict the reactivity of elements and design new materials with desired properties. This understanding is vital in various fields:
- Material Science: Designing new materials with specific properties.
- Pharmacology: Developing new drugs and understanding their interactions with the body.
- Environmental Science: Understanding the behavior of elements in the environment and their impact on ecosystems.
- Nuclear Chemistry: Studying the properties and applications of radioactive elements.
Conclusion: Groups – The Key to Understanding Chemical Behavior
In conclusion, the columns on the periodic table, known as groups or families, are essential for understanding the chemical properties of elements. The number of valence electrons, directly linked to the group number, dictates an element's reactivity and the types of compounds it forms. By studying the trends and properties of elements within each group, we can unlock a deeper understanding of chemical behavior and pave the way for advancements in various scientific and technological fields. From the highly reactive alkali metals to the inert noble gases, each group holds unique properties and applications, making the periodic table a powerful tool for chemical understanding and innovation. The continued exploration and study of groups within the periodic table remain crucial for advancing our knowledge of chemistry and its applications across various disciplines.
Latest Posts
Latest Posts
-
Labeling Exercise 8 2 Requisition And Blood Specimen Label
Apr 04, 2025
-
Which Are Qualities Of Abusive Conduct
Apr 04, 2025
-
Which Nims Command And Coordination Structures Are Offsite Locations
Apr 04, 2025
-
The Goal Of Sales Promotion Is To
Apr 04, 2025
-
An Abdominal Hernia Results When Weakened Muscles
Apr 04, 2025
Related Post
Thank you for visiting our website which covers about Columns On The Periodic Table Are Called . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
