Classify Each Of The Following As Acidic Basic Or Neutral
Holbox
Mar 31, 2025 · 6 min read
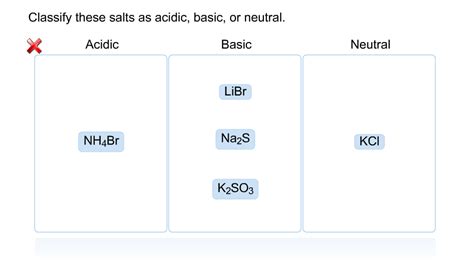
Table of Contents
- Classify Each Of The Following As Acidic Basic Or Neutral
- Table of Contents
- Classify Each of the Following as Acidic, Basic, or Neutral: A Comprehensive Guide
- Understanding the pH Scale
- The Role of Hydrogen and Hydroxide Ions
- Classifying Substances: A Detailed Approach
- 1. Using a pH Meter
- 2. Using pH Indicators
- 3. Understanding Chemical Formulas
- Examples of Acidic, Basic, and Neutral Substances
- The Importance of pH in Different Contexts
- 1. Biology and Medicine:
- 2. Environmental Science:
- 3. Industry:
- Conclusion
- Latest Posts
- Latest Posts
- Related Post
Classify Each of the Following as Acidic, Basic, or Neutral: A Comprehensive Guide
Understanding the pH scale and classifying substances as acidic, basic, or neutral is fundamental to numerous fields, from chemistry and biology to environmental science and everyday life. This comprehensive guide delves into the intricacies of pH, providing a detailed explanation of how to classify various substances and highlighting the importance of pH in different contexts. We'll explore various methods for determining pH and the implications of acidity and basicity in diverse applications.
Understanding the pH Scale
The pH scale is a logarithmic scale used to specify the acidity or basicity (alkalinity) of an aqueous solution. It ranges from 0 to 14, with 7 representing neutrality. Solutions with a pH less than 7 are acidic, while solutions with a pH greater than 7 are basic (alkaline). The scale is logarithmic, meaning each whole number change represents a tenfold change in hydrogen ion concentration ([H⁺]). For instance, a solution with a pH of 3 is ten times more acidic than a solution with a pH of 4, and one hundred times more acidic than a solution with a pH of 5.
The Role of Hydrogen and Hydroxide Ions
The acidity or basicity of a solution is determined by the relative concentrations of hydrogen ions (H⁺) and hydroxide ions (OH⁻). In pure water, the concentrations of these ions are equal, resulting in a neutral pH of 7. Acids increase the concentration of H⁺ ions, while bases increase the concentration of OH⁻ ions. The relationship between these ions is inverse; as the concentration of one increases, the concentration of the other decreases.
Classifying Substances: A Detailed Approach
Classifying a substance as acidic, basic, or neutral requires understanding its chemical properties and the resulting impact on the concentration of H⁺ and OH⁻ ions in solution. Let's examine several common methods and examples:
1. Using a pH Meter
A pH meter is an electronic instrument that directly measures the pH of a solution. It's a highly accurate method, often used in laboratories and industrial settings for precise pH determination. The meter employs a special electrode that responds to the concentration of hydrogen ions in the solution. The reading is displayed digitally, providing a precise pH value.
2. Using pH Indicators
pH indicators are substances that change color depending on the pH of the solution. These are often used for quick, less precise pH measurements. Litmus paper, a common indicator, turns red in acidic solutions and blue in basic solutions. Other indicators, such as phenolphthalein and methyl orange, exhibit different color changes over specific pH ranges, allowing for a more nuanced assessment of acidity or basicity. The color change is a visual indication of the pH, although it may not be as accurate as a pH meter.
3. Understanding Chemical Formulas
Many substances can be classified as acidic or basic simply by examining their chemical formulas.
-
Acids: Often contain hydrogen (H⁺) ions that dissociate (separate) in water, increasing the concentration of H⁺ ions. Examples include hydrochloric acid (HCl), sulfuric acid (H₂SO₄), and acetic acid (CH₃COOH). These acids are often referred to as strong acids (completely dissociate in water) or weak acids (partially dissociate).
-
Bases: Often contain hydroxide ions (OH⁻) which directly increase the hydroxide ion concentration in solution. Examples include sodium hydroxide (NaOH), potassium hydroxide (KOH), and calcium hydroxide (Ca(OH)₂). Some bases, however, don't directly contain hydroxide ions but react with water to produce them (e.g., ammonia, NH₃). These bases are often called strong bases (completely dissociate) or weak bases (partially dissociate).
-
Neutral Substances: These substances do not significantly alter the concentration of H⁺ or OH⁻ ions when dissolved in water. Pure water itself is a neutral substance, with a pH of 7. Many salts, such as sodium chloride (NaCl), also exhibit neutral pH when dissolved.
Examples of Acidic, Basic, and Neutral Substances
Let's classify some common substances based on their pH:
Acidic Substances (pH < 7):
- Lemon juice: Highly acidic due to the presence of citric acid. pH typically ranges from 2 to 3.
- Vinegar: Contains acetic acid, giving it a mildly acidic pH (around 3).
- Stomach acid: Primarily hydrochloric acid, essential for digestion, with a very low pH (around 1.5-3.5).
- Battery acid: Sulfuric acid, a highly corrosive strong acid with an extremely low pH.
- Cola drinks: Contain phosphoric acid and carbonic acid, making them acidic (pH around 2.5-3.5).
- Orange juice: Citric acid contributes to its acidic nature (pH around 3-4).
Basic Substances (pH > 7):
- Baking soda (sodium bicarbonate): A weak base, often used in baking and as an antacid.
- Ammonia: A weak base used in cleaning products.
- Bleach: A strong base containing sodium hypochlorite, used for disinfection.
- Soapy water: Soap increases the pH, making water slightly basic.
- Milk of Magnesia: Magnesium hydroxide suspension, used as a laxative and antacid.
- Lye (sodium hydroxide): A strong base, highly corrosive and dangerous if not handled correctly.
Neutral Substances (pH ≈ 7):
- Pure water: Under standard conditions, it has a pH of 7.
- Table salt (sodium chloride): A neutral salt when dissolved in water.
- Sugar (sucrose): A neutral organic compound.
The Importance of pH in Different Contexts
The pH of a solution significantly impacts various processes and systems. Understanding and controlling pH is crucial in numerous applications:
1. Biology and Medicine:
- Maintaining blood pH: Human blood must maintain a narrow pH range (7.35-7.45) for proper physiological function. Deviations from this range can lead to serious health consequences (acidosis or alkalosis).
- Enzyme activity: Enzymes, biological catalysts, function optimally within a specific pH range. Changes in pH can alter their shape and activity.
- Drug delivery: The pH of a solution can affect drug absorption and efficacy.
2. Environmental Science:
- Water quality: The pH of water is a critical indicator of water quality. Acid rain, for example, has a significantly lower pH than normal rainwater, causing harm to aquatic life and ecosystems.
- Soil pH: The pH of soil affects the availability of nutrients to plants. Different plants thrive in different pH ranges.
3. Industry:
- Chemical processes: Many chemical reactions are highly pH-dependent. Careful control of pH is often essential for efficient and effective reactions.
- Food preservation: pH control is critical in food preservation, as many microorganisms thrive within specific pH ranges.
- Water treatment: Adjusting the pH of water is a crucial step in water purification and treatment processes.
Conclusion
Classifying substances as acidic, basic, or neutral is essential for understanding a wide range of chemical and biological phenomena. Utilizing pH meters, indicators, or knowledge of chemical formulas allows for accurate determination of pH. The pH scale serves as a fundamental tool in diverse fields, highlighting the critical role of pH in maintaining balance and optimizing various processes. The knowledge and application of pH classification are not only relevant to scientific studies but are integral for ensuring safety, optimizing processes, and preserving the balance of our environment. From everyday applications to complex industrial processes, a solid understanding of pH is indispensable.
Latest Posts
Latest Posts
-
What Is The Equivalent Resistance Between Points A And B
Apr 03, 2025
-
A Customer Is Traveling To A Branch Office
Apr 03, 2025
-
Setting Up The Math For A Two Step Quantitative Problem
Apr 03, 2025
-
What Key Role Does An Investment Bank Play
Apr 03, 2025
-
Which Bacterial Strain Is The Least Competitively Dominant
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Classify Each Of The Following As Acidic Basic Or Neutral . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
