All Information Recorded On The Pcr Must Be
Holbox
Apr 02, 2025 · 7 min read
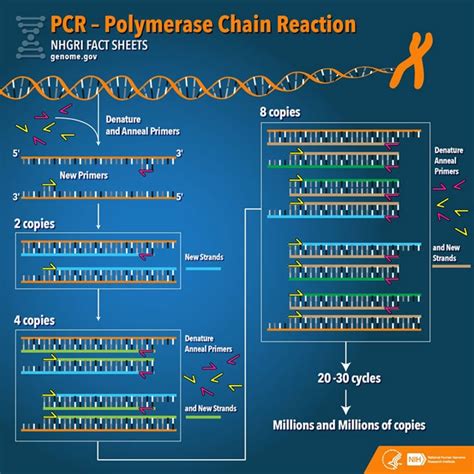
Table of Contents
- All Information Recorded On The Pcr Must Be
- Table of Contents
- All Information Recorded on the PCR Must Be: A Comprehensive Guide to PCR Data Integrity
- Pre-PCR Phase: Laying the Foundation for Reliable Results
- 1. Experimental Design and Rationale:
- PCR Reaction and Amplification: Maintaining Rigorous Standards
- 2. PCR Reaction Setup and Parameters:
- Post-PCR Analysis and Interpretation: Ensuring Data Integrity
- 3. Data Analysis and Interpretation:
- Beyond the Basics: Addressing Advanced PCR Techniques and Considerations
- 4. Advanced PCR Techniques:
- Data Management and Storage: Ensuring Long-Term Accessibility and Integrity
- 5. Data Management and Storage:
- Conclusion: The Importance of PCR Data Integrity
- Latest Posts
- Latest Posts
- Related Post
All Information Recorded on the PCR Must Be: A Comprehensive Guide to PCR Data Integrity
The polymerase chain reaction (PCR) has revolutionized molecular biology, enabling scientists to amplify specific DNA sequences exponentially. This powerful technique is used extensively in various fields, from medical diagnostics to forensic science and environmental monitoring. However, the reliability and validity of PCR results hinge critically on the meticulous recording and management of all associated information. This comprehensive guide delves into the crucial data points that must be documented during every stage of a PCR experiment to ensure data integrity, reproducibility, and the overall trustworthiness of the findings.
Pre-PCR Phase: Laying the Foundation for Reliable Results
The success of a PCR experiment begins long before the thermal cycler is even switched on. Meticulous record-keeping during the pre-PCR phase is essential to establishing a robust and traceable experimental process. The following information must be documented:
1. Experimental Design and Rationale:
- Detailed description of the research question or hypothesis: Why is this PCR being performed? What specific information is sought? A clear articulation of the experimental objectives is paramount.
- Target gene/sequence information: Precise details of the target DNA sequence, including its GenBank accession number (if applicable), its location within the genome, and its expected size.
- Primer design and sequences: Include the exact nucleotide sequences of all forward and reverse primers used, their annealing temperatures (Tm), and their GC content. Document the software used for primer design and any modifications made. Justify the primer choice, explaining why these specific primers were selected. Mentioning potential off-target binding sites, if any, is critical for transparency.
- Expected amplicon size: Accurately calculate the expected size of the PCR product based on the primer sequences and their location on the target DNA. This is crucial for accurate gel electrophoresis analysis.
- Positive and negative controls: Clearly specify the nature and source of positive and negative controls employed. Positive controls should yield the expected amplicon, while negative controls (e.g., no template control) should not show amplification. These controls are essential to assess the specificity and reliability of the PCR reaction.
- Sample preparation methods: A detailed protocol for DNA extraction, purification, and quantification must be documented. This includes the type of kit used (if applicable), the specific steps followed, and the concentration and purity of the extracted DNA. Mention any quality control measures performed (e.g., spectrophotometry readings, gel electrophoresis).
- Master mix components and concentrations: Precisely record all components of the master mix (e.g., DNA polymerase, dNTPs, MgCl2, buffer), including their concentrations and suppliers. The batch numbers of reagents should also be recorded for future traceability.
PCR Reaction and Amplification: Maintaining Rigorous Standards
The PCR reaction itself is a critical stage where errors can easily be introduced if procedures aren’t strictly adhered to and meticulously recorded.
2. PCR Reaction Setup and Parameters:
- Sample and reagent volumes: Record the exact volume of each component added to each PCR reaction tube, including the template DNA, master mix, and primers. This should be consistent across all reactions.
- Thermal cycling conditions: Detail the PCR thermocycling parameters used (e.g., denaturation temperature, annealing temperature, extension temperature, number of cycles). Justify the chosen parameters, explaining why they are appropriate for the specific target sequence and primers used.
- Thermal cycler model and software version: Specify the model of the thermal cycler used, including its software version. This is crucial for reproducibility, as different thermal cyclers might have slight variations in temperature control.
- Date and time of the PCR run: Note the exact date and time when the PCR was initiated. This provides a chronological record of the experiment.
Post-PCR Analysis and Interpretation: Ensuring Data Integrity
Post-PCR analysis is equally critical in ensuring the reliability and validity of the results.
3. Data Analysis and Interpretation:
- Gel electrophoresis results: Record the size of the amplified DNA fragments (if applicable) visualized on the gel. Include information such as the percentage of agarose used, the running voltage and time, the staining method employed (e.g., ethidium bromide, SYBR Safe), and the inclusion of a DNA ladder. Take high-quality photographs or scans of the gel and store them with the data. Quantify the band intensities whenever possible to allow for comparisons between samples.
- Quantification methods (qPCR): If quantitative PCR (qPCR) was performed, detail the method used (e.g., absolute quantification, relative quantification), the reference gene(s), the calibration curves, and the Ct values obtained for each sample. Describe the statistical analysis methods employed to determine significant differences between groups.
- Sequencing results: If DNA sequencing was performed, include the raw sequencing data (.ab1 files), the sequence chromatograms, and the final aligned consensus sequences. Clearly identify any ambiguities or low-quality regions in the sequence. Analyze the sequences using appropriate bioinformatics tools and report any mutations or variations observed.
- Data analysis software: Specify the names and versions of all software used for data analysis. This ensures transparency and allows others to reproduce the analysis.
Beyond the Basics: Addressing Advanced PCR Techniques and Considerations
The meticulous record-keeping described above applies to basic PCR techniques. However, additional considerations are necessary for advanced PCR methods.
4. Advanced PCR Techniques:
- Nested PCR: If nested PCR was used, document the primer sequences for both the primary and nested PCR reactions. Clearly indicate the cycling conditions for each reaction. The rationale behind using nested PCR should also be clearly stated.
- Real-time PCR (qPCR): For qPCR, additional data should be recorded, such as the fluorescence readings at each cycle, the melting curve analysis, and the calculation of the efficiency of the PCR reaction. Include a detailed description of the normalization strategy if relative quantification was employed.
- Reverse transcription PCR (RT-PCR): If RT-PCR was used for RNA amplification, the method of reverse transcription should be documented, including the type of reverse transcriptase used and the conditions of the reaction. The quality of the RNA template should also be assessed before starting the RT-PCR.
- Digital PCR (dPCR): For dPCR, the specific parameters of the partitioning process should be recorded, including the number of partitions and the distribution of positive and negative partitions. The analysis method used for determining the concentration of the target DNA should also be documented.
Data Management and Storage: Ensuring Long-Term Accessibility and Integrity
5. Data Management and Storage:
- Data storage format: All data, including raw data files, analysis results, and laboratory notebooks, should be stored in a structured and well-organized manner. Use appropriate file naming conventions to ensure clarity and easy retrieval.
- Data backup and security: Implement a reliable system for backing up all data to prevent data loss due to hardware failure or other unforeseen events. Secure data access and limit it to authorized personnel.
- Data version control: Use version control systems to track changes in data files and analysis scripts. This allows for easy tracking of modifications and ensures that the most up-to-date version is always available.
- Metadata: Each data file should be accompanied by detailed metadata, providing context and facilitating data interpretation. Metadata should include information such as the experiment date, the experimenter's name, the sample source, and the experimental conditions.
- Data sharing and publication: When sharing data with collaborators or publishing results, consider using standardized data formats and repositories to ensure data accessibility and reproducibility.
Conclusion: The Importance of PCR Data Integrity
The information recorded during a PCR experiment goes far beyond simple experimental parameters. It encompasses a comprehensive narrative of the entire process, from initial hypothesis to final data interpretation. This detailed record is crucial for data validation, result reproducibility, and ultimately, the credibility of scientific findings. By adhering to these guidelines, researchers can ensure the quality, reliability, and long-term value of their PCR data, contributing to a more robust and trustworthy scientific landscape. The meticulous documentation of every step guarantees transparency and enables others to verify the findings, furthering the advancement of scientific knowledge. Failing to record the essential details compromises the scientific rigor and casts doubt upon the validity of the conclusions. Therefore, maintaining complete and accurate PCR records is not merely a good practice; it is an essential aspect of responsible scientific research.
Latest Posts
Latest Posts
-
Asphalt At 120 F Considered To Be A Newtonian
Apr 05, 2025
-
Low Voltage Power Circuit Breakers Are Listed Under Ul Standard
Apr 05, 2025
-
Derek Wants To Win The Student Of The Year
Apr 05, 2025
-
Speciation Is Best Described As The
Apr 05, 2025
-
Explain What Is Misleading About The Graphic
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about All Information Recorded On The Pcr Must Be . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
