1 3 Butadiene Undergoes An Electrophilic Addition With Hbr
Holbox
Mar 30, 2025 · 5 min read
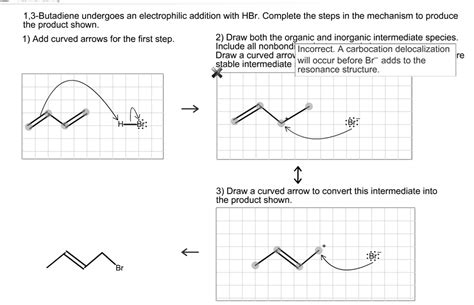
Table of Contents
- 1 3 Butadiene Undergoes An Electrophilic Addition With Hbr
- Table of Contents
- 1,3-Butadiene: Electrophilic Addition with HBr – A Deep Dive
- The Mechanism of Electrophilic Addition: A Step-by-Step Approach
- Step 1: Formation of the Allylic Carbocation
- Step 2: Nucleophilic Attack and Product Formation
- Regioselectivity: 1,2- vs. 1,4-Addition Products
- Stereochemistry: Cis and Trans Isomers
- Influence of Reaction Conditions
- Applications and Significance
- Conclusion: A Complex Reaction with Diverse Outcomes
- Latest Posts
- Latest Posts
- Related Post
1,3-Butadiene: Electrophilic Addition with HBr – A Deep Dive
1,3-Butadiene, a conjugated diene, exhibits unique reactivity compared to isolated dienes due to its extended π-electron system. This article delves into the electrophilic addition reaction of HBr with 1,3-butadiene, exploring the mechanism, regioselectivity, stereochemistry, and the influence of reaction conditions. We'll also examine the different products formed and the factors that govern their relative yields. Understanding this reaction is crucial for comprehending the reactivity of conjugated systems and their applications in organic synthesis.
The Mechanism of Electrophilic Addition: A Step-by-Step Approach
The reaction between 1,3-butadiene and HBr proceeds via an electrophilic addition mechanism. Unlike simple alkenes, the reaction with a conjugated diene offers a unique pathway, leading to a mixture of products. The mechanism involves two steps:
Step 1: Formation of the Allylic Carbocation
The reaction initiates with the electrophilic attack of the proton (H⁺) from HBr on one of the double bonds in 1,3-butadiene. Because of the conjugation, the positive charge is delocalized over two carbon atoms, forming an allylic carbocation intermediate. This is a crucial step distinguishing this reaction from the addition to isolated alkenes. The delocalization stabilizes the carbocation, making the reaction more favorable. The resonance structures of this intermediate are shown below:
(Insert image here: Resonance structures of the allylic carbocation intermediate. Show the positive charge delocalized over carbons 1 and 3.)
This allylic carbocation is significantly more stable than a simple secondary carbocation, explaining the enhanced reactivity of conjugated dienes compared to isolated alkenes. The stability arises from the resonance stabilization, which distributes the positive charge over multiple atoms.
Step 2: Nucleophilic Attack and Product Formation
In the second step, the bromide ion (Br⁻) acts as a nucleophile, attacking either carbon 1 or carbon 3 of the allylic carbocation. Attack at carbon 1 yields 3-bromo-1-butene (the 1,2-addition product), while attack at carbon 3 leads to 1-bromo-2-butene (the 1,4-addition product). Both products are possible due to the resonance-stabilized nature of the intermediate carbocation.
(Insert image here: Mechanism showing the nucleophilic attack by Br⁻ on both carbons of the allylic carbocation, leading to 1,2- and 1,4-addition products.)
Regioselectivity: 1,2- vs. 1,4-Addition Products
The reaction's regioselectivity—the preference for one regioisomer over another—is temperature-dependent. At lower temperatures (typically below -80°C), the 1,2-addition product (3-bromo-1-butene) predominates. This is because the kinetic control favors the faster reaction, which involves the formation of the more substituted (but less stable) carbocation intermediate. The nucleophile attacks the carbocation immediately upon formation, leading primarily to the 1,2-addition product.
At higher temperatures (typically above 0°C), the thermodynamic control takes over, and the 1,4-addition product (1-bromo-2-butene) becomes the major product. At higher temperatures, the carbocation intermediate has sufficient time to equilibrate between its resonance forms. The more stable product, 1-bromo-2-butene (due to greater substitution and conjugation), is formed predominantly because it has a lower overall energy.
(Insert image here: Energy diagram showing the kinetic and thermodynamic control of the 1,2- and 1,4-addition products. Label the activation energies and relative energies of the products.)
Stereochemistry: Cis and Trans Isomers
The 1,4-addition product, 1-bromo-2-butene, can exist as both cis and trans isomers. The stereochemistry of the product depends on the approach of the bromide ion to the allylic carbocation. The trans isomer is generally favored due to steric factors, minimizing interactions between the substituents on the double bond. However, a mixture of cis and trans isomers is typically observed.
(Insert image here: Structures of cis and trans-1-bromo-2-butene.)
The 1,2-addition product, 3-bromo-1-butene, does not exhibit cis-trans isomerism as it only has one double bond and no stereoisomers are possible in this case.
Influence of Reaction Conditions
Several factors influence the outcome of the electrophilic addition of HBr to 1,3-butadiene:
-
Temperature: As discussed above, temperature plays a critical role in determining the regioselectivity of the reaction, favoring 1,2-addition at low temperatures and 1,4-addition at high temperatures.
-
Solvent: The choice of solvent can affect the reaction rate and selectivity. Polar solvents can stabilize the carbocation intermediate, influencing the reaction pathway.
-
Concentration of Reactants: The relative concentrations of 1,3-butadiene and HBr can also affect the product distribution.
Applications and Significance
The electrophilic addition of HBr to 1,3-butadiene is not just an academic exercise. It showcases the fundamental principles of electrophilic addition to conjugated systems, providing insight into the reaction mechanisms and the factors governing product selectivity. This understanding is invaluable in designing and predicting the outcome of various organic reactions involving conjugated dienes. The products obtained from this reaction can serve as important intermediates in the synthesis of more complex organic molecules, finding applications in various fields including polymer chemistry, pharmaceutical synthesis and materials science. For example, the resulting bromoalkenes can undergo further reactions such as substitution or elimination to yield a variety of useful compounds.
Conclusion: A Complex Reaction with Diverse Outcomes
The electrophilic addition of HBr to 1,3-butadiene is a rich and complex reaction, exhibiting both regioselectivity and stereochemistry. The temperature dependence, along with the influence of resonance stabilization of the intermediate carbocation, dictates the final product distribution. Understanding this reaction provides critical insights into the behavior of conjugated dienes and their importance in organic chemistry. This knowledge is fundamental for organic chemists engaged in synthesis and for comprehending the intricate reactivity of organic molecules. The ability to control the reaction conditions and predict the product ratios is key to achieving desired outcomes in various applications. Further research continues to refine our understanding of this fundamental reaction and its applications in various fields. The detailed understanding of this reaction lays the foundation for exploring more complex electrophilic additions in conjugated systems and designing efficient synthetic strategies.
Latest Posts
Latest Posts
-
Which Of The Following Is An Example Of Operant Conditioning
Apr 03, 2025
-
Olfaction And Hearing Are Processed In The
Apr 03, 2025
-
Economists Have Difficulty Applying The Scientific Method Because
Apr 03, 2025
-
Criminal Justice In America 10th Edition
Apr 03, 2025
-
A Companys Strategy Is At Full Power
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about 1 3 Butadiene Undergoes An Electrophilic Addition With Hbr . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
